 |
PDBsum entry 2duc

|

|

|
|
 |
Contents |
 |
|
|

|

|
|
|
|
|
|
* Residue conservation analysis
|
|
|

|
|
PDB id:
|
 |
|
 |
| Name: |
 |
Hydrolase
|
 |
|
Title:
|
 |
Crystal structure of sars coronavirus main proteinase(3clpro)
|
|
Structure:
|
 |
Replicase polyprotein 1ab. Chain: a, b. Fragment: 3c-like proteinase. Synonym: 3cl-pro, 3clp, nsp2. Engineered: yes
|
|
Source:
|
 |
Sars coronavirus. Organism_taxid: 227859. Other_details: cell-free protein synthesis
|
|
Resolution:
|
 |
|
1.70Å
|
R-factor:
|
0.194
|
R-free:
|
0.221
|
|
|
Authors:
|
 |
H.Wang,Y.T.Kim,T.Muramatsu,C.Takemoto,M.Shirouzu,S.Yokoyama,Riken Structural Genomics/proteomics Initiative (Rsgi)
|
|
Key ref:
|
 |
T.Muramatsu
et al.
(2016).
SARS-CoV 3CL protease cleaves its C-terminal autoprocessing site by novel subsite cooperativity.
Proc Natl Acad Sci U S A,
113,
12997-13002.
PubMed id:
|
 |
|
Date:
|
 |
|
21-Jul-06
|
Release date:
|
24-Jul-07
|
|
|

|

|
|
PROCHECK
|

|

|
|
|
Headers
|
 |
|
|
References
|
|
|
|

|

|
|
P0C6X7
(R1AB_CVHSA) -
Replicase polyprotein 1ab from Severe acute respiratory syndrome coronavirus
|
|

|

|
Seq:
Struc:
|
 |
 |
 |
7073 a.a.
306 a.a.
|

|

|

|

|
|

|

|

|
 |
 |

|
|
Key: |
 |
PfamA domain |
 |
 |
 |
Secondary structure |
 |
 |
CATH domain |
 |
|

|
|
|
 |
|
|
 |
 |
 |
 |
Enzyme class 2:
|
 |
E.C.2.1.1.-
- ?????
|
|
 |
 |
 |
 |
 |
Enzyme class 3:
|
 |
E.C.2.1.1.56
- mRNA (guanine-N(7))-methyltransferase.
|
|
 |
 |
 |
 |
 |
Reaction:
|
 |
a 5'-end (5'-triphosphoguanosine)-ribonucleoside in mRNA + S-adenosyl-L- methionine = a 5'-end (N(7)-methyl 5'-triphosphoguanosine)-ribonucleoside in mRNA + S-adenosyl-L-homocysteine
|
 |
 |
 |
 |
 |
5'-end (5'-triphosphoguanosine)-ribonucleoside in mRNA
|
+
|
 S-adenosyl-L- methionine
S-adenosyl-L- methionine
|
=
|
5'-end (N(7)-methyl 5'-triphosphoguanosine)-ribonucleoside in mRNA
|
+
|
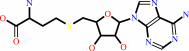 S-adenosyl-L-homocysteine
S-adenosyl-L-homocysteine
|
|
 |
 |
 |
 |
 |
 |
 |
 |
Enzyme class 4:
|
 |
E.C.2.1.1.57
- methyltransferase cap1.
|
|
 |
 |
 |
 |
 |
Reaction:
|
 |
a 5'-end (N(7)-methyl 5'-triphosphoguanosine)-ribonucleoside in mRNA + S-adenosyl-L-methionine = a 5'-end (N(7)-methyl 5'-triphosphoguanosine)- (2'-O-methyl-ribonucleoside) in mRNA + S-adenosyl-L-homocysteine + H+
|
 |
 |
 |
 |
 |
5'-end (N(7)-methyl 5'-triphosphoguanosine)-ribonucleoside in mRNA
|
+
|
 S-adenosyl-L-methionine
S-adenosyl-L-methionine
|
=
|
5'-end (N(7)-methyl 5'-triphosphoguanosine)- (2'-O-methyl-ribonucleoside) in mRNA
|
+
|
 S-adenosyl-L-homocysteine
S-adenosyl-L-homocysteine
|
+
|
H(+)
|
|
 |
 |
 |
 |
 |
 |
 |
 |
Enzyme class 5:
|
 |
E.C.2.7.7.48
- RNA-directed Rna polymerase.
|
|
 |
 |
 |
 |
 |
Reaction:
|
 |
RNA(n) + a ribonucleoside 5'-triphosphate = RNA(n+1) + diphosphate
|
 |
 |
 |
 |
 |
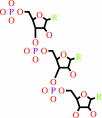 RNA(n)
RNA(n)
|
+
|
ribonucleoside 5'-triphosphate
|
=
|
RNA(n+1)
|
+
|
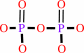 diphosphate
diphosphate
|
|
 |
 |
 |
 |
 |
 |
 |
 |
Enzyme class 6:
|
 |
E.C.2.7.7.50
- mRNA guanylyltransferase.
|
|
 |
 |
 |
 |
 |
Reaction:
|
 |
a 5'-end diphospho-ribonucleoside in mRNA + GTP + H+ = a 5'-end (5'-triphosphoguanosine)-ribonucleoside in mRNA + diphosphate
|
 |
 |
 |
 |
 |
5'-end diphospho-ribonucleoside in mRNA
|
+
|
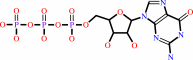 GTP
GTP
|
+
|
H(+)
|
=
|
5'-end (5'-triphosphoguanosine)-ribonucleoside in mRNA
|
+
|
 diphosphate
diphosphate
|
|
 |
 |
 |
 |
 |
 |
 |
 |
Enzyme class 7:
|
 |
E.C.3.1.13.-
- ?????
|
|
 |
 |
 |
 |
 |
Enzyme class 8:
|
 |
E.C.3.4.19.12
- ubiquitinyl hydrolase 1.
|
|
 |
 |
 |
 |
 |
Reaction:
|
 |
Thiol-dependent hydrolysis of ester, thiolester, amide, peptide and isopeptide bonds formed by the C-terminal Gly of ubiquitin (a 76-residue protein attached to proteins as an intracellular targeting signal).
|
 |
 |
 |
 |
 |
Enzyme class 9:
|
 |
E.C.3.4.22.-
- ?????
|
|
 |
 |
 |
 |
 |
Enzyme class 10:
|
 |
E.C.3.4.22.69
- Sars coronavirus main proteinase.
|
|
 |
 |
 |
 |
 |
Enzyme class 11:
|
 |
E.C.3.6.4.12
- Dna helicase.
|
|
 |
 |
 |
 |
 |
Reaction:
|
 |
ATP + H2O = ADP + phosphate + H+
|
 |
 |
 |
 |
 |
 ATP
ATP
|
+
|
H2O
|
=
|
 ADP
ADP
|
+
|
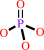 phosphate
phosphate
|
+
|
H(+)
|
|
 |
 |
 |
 |
 |
 |
 |
 |
Enzyme class 12:
|
 |
E.C.3.6.4.13
- Rna helicase.
|
|
 |
 |
 |
 |
 |
Reaction:
|
 |
ATP + H2O = ADP + phosphate + H+
|
 |
 |
 |
 |
 |
 ATP
ATP
|
+
|
H2O
|
=
|
 ADP
ADP
|
+
|
 phosphate
phosphate
|
+
|
H(+)
|
|
 |
 |
 |
 |
 |
 |
 |
 |
Enzyme class 13:
|
 |
E.C.4.6.1.-
- ?????
|
|
 |
 |
 |
 |
 |
 |
 |
|
Note, where more than one E.C. class is given (as above), each may
correspond to a different protein domain or, in the case of polyprotein
precursors, to a different mature protein.
|
|
 |
|
Molecule diagrams generated from .mol files obtained from the
KEGG ftp site
|
|
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |

|

|

|
| |
|
|
| |
|
|
Proc Natl Acad Sci U S A
113:12997-13002
(2016)
|
|
PubMed id:
|
|
|
|

|
| |
|
SARS-CoV 3CL protease cleaves its C-terminal autoprocessing site by novel subsite cooperativity.
|
|
T.Muramatsu,
C.Takemoto,
Y.T.Kim,
H.Wang,
W.Nishii,
T.Terada,
M.Shirouzu,
S.Yokoyama.
|
|
|

|
| |
ABSTRACT
|
|

|
| |

|
The 3C-like protease (3CLpro) of severe acute respiratory syndrome
coronavirus (SARS-CoV) cleaves 11 sites in the polyproteins, including its own
N- and C-terminal autoprocessing sites, by recognizing P4-P1 and P1'. In this
study, we determined the crystal structure of 3CLprowith the
C-terminal prosequence and the catalytic-site C145A mutation, in which the
enzyme binds the C-terminal prosequence of another molecule. Surprisingly, Phe
at the P3' position [Phe(P3')] is snugly accommodated in the S3' pocket.
Mutations of Phe(P3') impaired the C-terminal autoprocessing, but did not affect
N-terminal autoprocessing. This difference was ascribed to the P2 residue,
Phe(P2) and Leu(P2), in the C- and N-terminal sites, as follows. The S3' subsite
is formed by Phe(P2)-induced conformational changes of 3CLproand the
direct involvement of Phe(P2) itself. In contrast, the N-terminal prosequence
with Leu(P2) does not cause such conformational changes for the S3' subsite
formation. In fact, the mutation of Phe(P2) to Leu in the C-terminal
autoprocessing site abolishes the dependence on Phe(P3'). These mechanisms
explain why Phe is required at the P3' position when the P2 position is occupied
by Phe rather than Leu, which reveals a type of subsite cooperativity. Moreover,
the peptide consisting of P4-P1 with Leu(P2) inhibits protease activity, whereas
that with Phe(P2) exhibits a much smaller inhibitory effect, because Phe(P3') is
missing. Thus, this subsite cooperativity likely exists to avoid the
autoinhibition of the enzyme by its mature C-terminal sequence, and to retain
the efficient C-terminal autoprocessing by the use of Phe(P2).

|

|
|
|

|

|
|
 |
 |
|
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |

