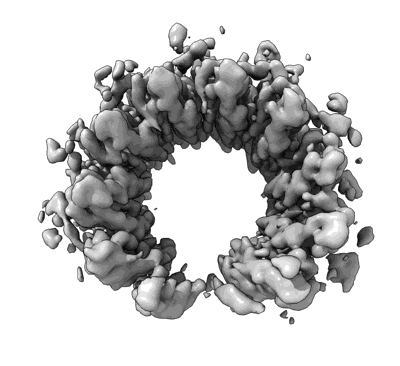Do data resources managed by EMBL-EBI and our collaborators make a difference to your work? Please take 10 minutes to fill in our annual user survey, and help us make the case for why sustaining open data resources is critical for life sciences research. We appreciate your responses prior to closing of the survey on Monday 17th June.
Resolution: 2.9 Å
EM Method: Single-particle
Fitted PDBs: 8ril
Q-score: 0.551
Human RAD52 open ring conformation
Resolution: 3.2 Å
EM Method: Single-particle
Fitted PDBs: 8rj3
Q-score: 0.325
Liang CC, Greenhough LA, Masino L, Maslen S, Bajrami I, Tuppi M, Skehel M, Taylor IA, West SC
Nature (2024) 629 pp. 697-703 [ DOI: doi:10.1038/s41586-024-07347-7 Pubmed: 38658755 ]
- Dna repair protein rad52 homolog (46 kDa, Protein from Homo sapiens)
- Open ring conformation of human rad52 (Complex from Homo sapiens)
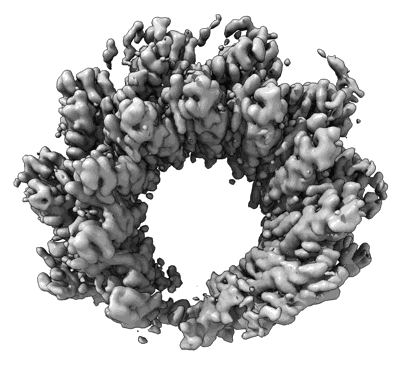
Human RAD52 open ring - ssDNA complex
Resolution: 2.3 Å
EM Method: Single-particle
Fitted PDBs: 8rjw
Q-score: 0.512
Liang CC, Greenhough LA, Masino L, Maslen S, Bajrami I, Tuppi M, Skehel M, Taylor IA, West SC
Nature (2024) 629 pp. 697-703 [ DOI: doi:10.1038/s41586-024-07347-7 Pubmed: 38658755 ]
- Open ring conformation of human rad52 in complex with ssdna (Complex from Homo sapiens)
- Magnesium ion (24 Da, Ligand)
- Water (18 Da, Ligand)
- Ssdna (6 kDa, DNA from Homo sapiens)
- Dna repair protein rad52 homolog (46 kDa, Protein from Homo sapiens)
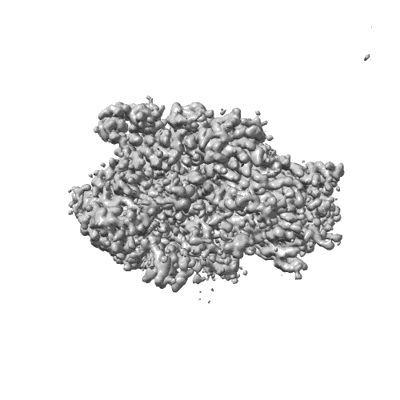
Human Replication protein A (RPA; trimeric core) - ssDNA complex
Resolution: 3.2 Å
EM Method: Single-particle
Fitted PDBs: 8rk2
Q-score: 0.436
Liang CC, Greenhough LA, Masino L, Maslen S, Bajrami I, Tuppi M, Skehel M, Taylor IA, West SC
Nature (2024) 629 pp. 697-703 [ DOI: doi:10.1038/s41586-024-07347-7 Pubmed: 38658755 ]
- Open ring conformation of human rad52 in complex with ssdna (Complex from Homo sapiens)
- Replication protein a 14 kda subunit (13 kDa, Protein from Homo sapiens)
- Replication protein a 70 kda dna-binding subunit, n-terminally processed (68 kDa, Protein from Homo sapiens)
- Replication protein a 32 kda subunit (29 kDa, Protein from Homo sapiens)
Structure and function of the RAD51B-RAD51C-RAD51D-XRCC2 tumour suppressor
Greenhough LA, Liang CC, Belan O, Kunzelmann S, Maslen S, Rodrigo-Brenni MC, Anand R, Skehel M, Boulton SJ, West SC
Nature (2023) 619 pp. 650-657 [ DOI: doi:10.1038/s41586-023-06179-1 Pubmed: 37344587 ]
Human RAD51B-RAD51C-RAD51D-XRCC2 (BCDX2) complex, 3.4 A resolution
Resolution: 3.4 Å
EM Method: Single-particle
Fitted PDBs: 8ouy
Q-score: 0.523
Greenhough LA, Liang CC, Belan O, Kunzelmann S, Maslen S, Rodrigo-Brenni MC, Anand R, Skehel M, Boulton SJ, West SC
Nature (2023) 619 pp. 650-657 [ DOI: doi:10.1038/s41586-023-06179-1 Pubmed: 37344587 ]
- Rad51b-rad51c-rad51d-xrcc2 (bcdx2) (Complex from Homo sapiens)
- Dna repair protein rad51 homolog 2 (38 kDa, Protein from Homo sapiens)
- Adenosine-5'-diphosphate (427 Da, Ligand)
- Dna repair protein xrcc2 (31 kDa, Protein from Homo sapiens)
- Adenosine-5'-triphosphate (507 Da, Ligand)
- Dna repair protein rad51 homolog 3 (42 kDa, Protein from Homo sapiens)
- Magnesium ion (24 Da, Ligand)
- Dna repair protein rad51 homolog 4 (35 kDa, Protein from Homo sapiens)
Human RAD51B-RAD51C-RAD51D-XRCC2 (BCDX2) complex, 2.2 A resolution
Resolution: 2.2 Å
EM Method: Single-particle
Fitted PDBs: 8ouz
Q-score: 0.625
Greenhough LA, Liang CC, Belan O, Kunzelmann S, Maslen S, Rodrigo-Brenni MC, Anand R, Skehel M, Boulton SJ, West SC
Nature (2023) 619 pp. 650-657 [ DOI: doi:10.1038/s41586-023-06179-1 Pubmed: 37344587 ]
- Dna repair protein xrcc2 (31 kDa, Protein from Homo sapiens)
- Rad51b-rad51c-rad51d-xrcc2 (bcdx2) (Complex from Homo sapiens)
- Dna repair protein rad51 homolog 2 (38 kDa, Protein from Homo sapiens)
- Adenosine-5'-diphosphate (427 Da, Ligand)
- Adenosine-5'-triphosphate (507 Da, Ligand)
- Dna repair protein rad51 homolog 3 (42 kDa, Protein from Homo sapiens)
- Magnesium ion (24 Da, Ligand)
- Water (18 Da, Ligand)
- Dna repair protein rad51 homolog 4 (35 kDa, Protein from Homo sapiens)
human DEPTOR in a complex with mutant human mTORC1 A1459P
Resolution: 4.7 Å
EM Method: Single-particle
Fitted PDBs: 7owg
Q-score: 0.293
Heimhalt M, Berndt A, Wagstaff J, Anandapadamanaban M, Perisic O, Maslen S, McLaughlin S, Yu CW, Masson GR, Boland A, Ni X, Yamashita K, Murshudov GN, Skehel M, Freund SM, Williams RL
eLife (2021) 10 [ Pubmed: 34519269 DOI: doi:10.7554/eLife.68799 ]
- Complex of human mutant mtorc1 a1459p with the inhibitory protein deptor (46 MDa, Complex)
- Mtorc1 with mutant a1459p mtor (Complex from Homo sapiens)
- Target of rapamycin complex subunit lst8 (35 kDa, Protein from Homo sapiens)
- Serine/threonine-protein kinase mtor (287 kDa, Protein from Homo sapiens)
- Regulatory-associated protein of mtor (149 kDa, Protein from Homo sapiens)
- Deptor (Complex from Homo sapiens)
- Dep domain-containing mtor-interacting protein (46 kDa, Protein from Homo sapiens)
Liu Y, Bunney TD, Khosa S, Mace K, Beckenbauer K, Askwith T, Maslen S, Stubbs C, de Oliveira TM, Sader K, Skehel M, Gavin AC, Phillips C, Katan M
Ebiomedicine (2020) 51 pp. 102607-102607 [ DOI: doi:10.1016/j.ebiom.2019.102607 Pubmed: 31918402 ]
- Protein complex (Complex from Homo sapiens)
Volume 1 for truncated dimeric Cytohesin-3 (Grp1; amino acids 14-399)
Resolution: 53.0 Å
EM Method: Single-particle
Fitted PDBs: 6u3e
Q-score: 0.043
Das S, Malaby AW, Nawrotek A, Zhang W, Zeghouf M, Maslen S, Skehel M, Chakravarthy S, Irving TC, Bilsel O, Cherfils J, Lambright DG
Structure (2019) 27 pp. 1782- [ Pubmed: 31601460 DOI: doi:10.1016/j.str.2019.09.007 ]