Do data resources managed by EMBL-EBI and our collaborators make a difference to your work? Please take 10 minutes to fill in our annual user survey, and help us make the case for why sustaining open data resources is critical for life sciences research. We appreciate your responses prior to closing of the survey on Monday 17th June.
Resolution: 3.06 Å
EM Method: Single-particle
Fitted PDBs: 8bei
Q-score: 0.543
Sendker LF, Lo YK, Heimerl T, Bohn S, Pfister P, Mais N, Sadowska W, Peterson L, Benesch JLP, Marklund E, Bange G, Erb TJ, Schuller JM, Hochberg GKA
To Be Published
Resolution: 5.91 Å
EM Method: Single-particle
Fitted PDBs: 8rjk
Q-score: 0.277
Sendker FL, Lo YK, Heimerl T, Bohn S, Persson LJ, Mais CN, Sadowska W, Paczia N, Nussbaum E, Del Carmen Sanchez Olmos M, Forchhammer K, Schindler D, Erb TJ, Benesch JLP, Marklund EG, Bange G, Schuller JM, Hochberg GKA
Nature (2024) 628 pp. 894-900 [ Pubmed: 38600380 DOI: doi:10.1038/s41586-024-07287-2 ]
Resolution: 3.34 Å
EM Method: Single-particle
Fitted PDBs: 8rjl
Q-score: 0.504
Sendker FL, Lo YK, Heimerl T, Bohn S, Persson LJ, Mais CN, Sadowska W, Paczia N, Nussbaum E, Del Carmen Sanchez Olmos M, Forchhammer K, Schindler D, Erb TJ, Benesch JLP, Marklund EG, Bange G, Schuller JM, Hochberg GKA
Nature (2024) 628 pp. 894-900 [ Pubmed: 38600380 DOI: doi:10.1038/s41586-024-07287-2 ]
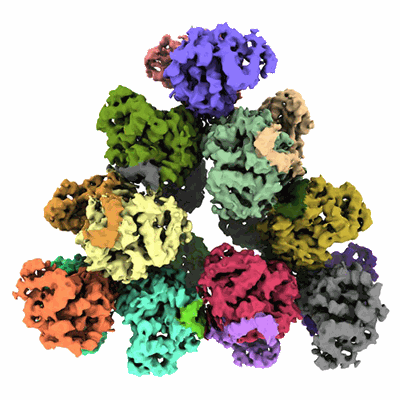
Structure of a first level Sierpinski triangle formed by a citrate synthase
Resolution: 3.93 Å
EM Method: Single-particle
Fitted PDBs: 8an1
Q-score: 0.386
Sendker LF, Lo YK, Heimerl T, Bohn S, Pfister P, Mais N, Sadowska W, Peterson L, Benesch JLP, Marklund E, Bange G, Erb TJ, Schuller JM, Hochberg GKA
To Be Published
Structure of ACLY-D1026A-substrates
Resolution: 2.2 Å
EM Method: Single-particle
Fitted PDBs: 7rig
Q-score: 0.588
Wei X, Schultz K, Pepper HL, Megill E, Vogt A, Snyder NW, Marmorstein R
Nat Commun (2023) 14 pp. 2247-2247 [ DOI: doi:10.1038/s41467-023-37986-9 Pubmed: 37076498 ]
- Coenzyme a (767 Da, Ligand)
- Citrate anion (189 Da, Ligand)
- Acly d1026a mutant in complex with coa and citrate (480 kDa, Complex from Homo sapiens)
- Atp-citrate synthase (120 kDa, Protein from Homo sapiens)
- Phosphate ion (94 Da, Ligand)
- Water (18 Da, Ligand)
- Adenosine-5'-diphosphate (427 Da, Ligand)
- Unknown ligand (189 Da, Ligand)
- (3s)-citryl-coenzyme a (941 Da, Ligand)
Structure of ACLY D1026A-substrates-asym-int
Resolution: 2.6 Å
EM Method: Single-particle
Fitted PDBs: 7rkz
Q-score: 0.412
Wei X, Schultz K, Pepper HL, Megill E, Vogt A, Snyder NW, Marmorstein R
Nat Commun (2023) 14 pp. 2247-2247 [ DOI: doi:10.1038/s41467-023-37986-9 Pubmed: 37076498 ]
- Coenzyme a (767 Da, Ligand)
- Unknown ligand (767 Da, Ligand)
- Citrate anion (189 Da, Ligand)
- Acly d1026a mutant in complex with coa and citrate (480 kDa, Complex from Homo sapiens)
- Atp-citrate synthase (120 kDa, Protein from Homo sapiens)
- Water (18 Da, Ligand)
- Adenosine-5'-diphosphate (427 Da, Ligand)
- (3s)-citryl-coenzyme a (941 Da, Ligand)
Structure of ACLY D1026A - substrates-asym
Resolution: 2.7 Å
EM Method: Single-particle
Fitted PDBs: 7rmp
Q-score: 0.427
Wei X, Schultz K, Pepper HL, Megill E, Vogt A, Snyder NW, Marmorstein R
Nat Commun (2023) 14 pp. 2247-2247 [ DOI: doi:10.1038/s41467-023-37986-9 Pubmed: 37076498 ]
- Coenzyme a (767 Da, Ligand)
- Unknown ligand (767 Da, Ligand)
- Citrate anion (189 Da, Ligand)
- Acly d1026a mutant in complex with coa and citrate (480 kDa, Complex from Homo sapiens)
- Atp-citrate synthase (120 kDa, Protein from Homo sapiens)
- Phosphate ion (94 Da, Ligand)
- Water (18 Da, Ligand)
- Adenosine-5'-diphosphate (427 Da, Ligand)
- (3s)-citryl-coenzyme a (941 Da, Ligand)
Structure of ACLY-D1026A-products-asym
Resolution: 2.8 Å
EM Method: Single-particle
Fitted PDBs: 8g1e
Q-score: 0.424
Wei X, Schultz K, Pepper HL, Megill E, Vogt A, Snyder NW, Marmorstein R
Nat Commun (2023) 14 pp. 2247-2247 [ Pubmed: 37076498 DOI: doi:10.1038/s41467-023-37986-9 ]
- (3s)-citryl-coenzyme a (941 Da, Ligand)
- Phosphate ion (94 Da, Ligand)
- Acly d1026a mutant in complex with coa and citrate (480 kDa, Complex from Homo sapiens)
- Adenosine-5'-diphosphate (427 Da, Ligand)
- Acetyl coenzyme *a (809 Da, Ligand)
- Water (18 Da, Ligand)
- Unknown ligand (Ligand)
- Oxaloacetate ion (131 Da, Ligand)
- Atp-citrate synthase (120 kDa, Protein from Homo sapiens)
Structure of ACLY-D1026A-products
Resolution: 2.4 Å
EM Method: Single-particle
Fitted PDBs: 8g1f
Q-score: 0.581
Wei X, Schultz K, Pepper HL, Megill E, Vogt A, Snyder NW, Marmorstein R
Nat Commun (2023) 14 pp. 2247-2247 [ Pubmed: 37076498 DOI: doi:10.1038/s41467-023-37986-9 ]
- (3s)-citryl-coenzyme a (941 Da, Ligand)
- Phosphate ion (94 Da, Ligand)
- Acly d1026a mutant in complex with products (480 kDa, Complex from Homo sapiens)
- Adenosine-5'-diphosphate (427 Da, Ligand)
- Acetyl coenzyme *a (809 Da, Ligand)
- Water (18 Da, Ligand)
- Unknown ligand (Ligand)
- Oxaloacetate ion (131 Da, Ligand)
- Atp-citrate synthase (120 kDa, Protein from Homo sapiens)
Structure of ACLY-D1026A-substrates, local refinement of ASH domain
Resolution: 2.2 Å
EM Method: Single-particle
Fitted PDBs: 8g5c
Q-score: 0.635
Wei X, Schultz K, Pepper HL, Megill E, Vogt A, Snyder NW, Marmorstein R
Nat Commun (2023) 14 pp. 2247-2247 [ Pubmed: 37076498 DOI: doi:10.1038/s41467-023-37986-9 ]
- Citrate anion (189 Da, Ligand)
- (3s)-citryl-coenzyme a (941 Da, Ligand)
- Phosphate ion (94 Da, Ligand)
- Adenosine-5'-diphosphate (427 Da, Ligand)
- Acly d1026a mutant in complex with coa and citrate (480 kDa, Complex from Homo sapiens)
- Coenzyme a (767 Da, Ligand)
- Water (18 Da, Ligand)
- Atp-citrate synthase (120 kDa, Protein from Homo sapiens)