 |
PDBsum entry 1jms

|

|

|
|
 |
Contents |
 |
|
|

|

|
|
|
|
|
|
|
|
* Residue conservation analysis
|
|
|
|
 |
|
|
 |
 |
 |
 |
Enzyme class 1:
|
 |
E.C.2.7.7.31
- Dna nucleotidylexotransferase.
|
|
 |
 |
 |
 |
 |
Reaction:
|
 |
DNA(n) + a 2'-deoxyribonucleoside 5'-triphosphate = DNA(n+1) + diphosphate
|
 |
 |
 |
 |
 |
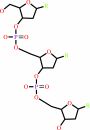 DNA(n)
DNA(n)
|
+
|
2'-deoxyribonucleoside 5'-triphosphate
|
=
|
DNA(n+1)
|
+
|
 diphosphate
diphosphate
|
|
 |
 |
 |
 |
 |
 |
 |
 |
Enzyme class 2:
|
 |
E.C.3.1.11.-
- ?????
|
|
 |
 |
 |
 |
 |
 |
 |
|
Note, where more than one E.C. class is given (as above), each may
correspond to a different protein domain or, in the case of polyprotein
precursors, to a different mature protein.
|
|
 |
|
Molecule diagrams generated from .mol files obtained from the
KEGG ftp site
|
|
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |

|

|

|
| |
|
|
| |
|
DOI no:
|
EMBO J
21:427-439
(2002)
|
|
PubMed id:
|
|
|
|

|
| |
|
Crystal structures of a template-independent DNA polymerase: murine terminal deoxynucleotidyltransferase.
|
|
M.Delarue,
J.B.Boulé,
J.Lescar,
N.Expert-Bezançon,
N.Jourdan,
N.Sukumar,
F.Rougeon,
C.Papanicolaou.
|
|
|

|
| |
ABSTRACT
|
|

|
| |

|
The crystal structure of the catalytic core of murine terminal
deoxynucleotidyltransferase (TdT) at 2.35 A resolution reveals a typical DNA
polymerase beta-like fold locked in a closed form. In addition, the structures
of two different binary complexes, one with an oligonucleotide primer and the
other with an incoming ddATP-Co(2+) complex, show that the substrates and the
two divalent ions in the catalytic site are positioned in TdT in a manner
similar to that described for the human DNA polymerase beta ternary complex,
suggesting a common two metal ions mechanism of nucleotidyl transfer in these
two proteins. The inability of TdT to accommodate a template strand can be
explained by steric hindrance at the catalytic site caused by a long lariat-like
loop, which is absent in DNA polymerase beta. However, displacement of this
discriminating loop would be sufficient to unmask a number of evolutionarily
conserved residues, which could then interact with a template DNA strand. The
present structure can be used to model the recently discovered human polymerase
mu, with which it shares 43% sequence identity.

|

|
|
|

|
| |
Selected figure(s)
|
|

|
| |
 |
 |

|
 |

|
 |
Figure 1.
Figure 1 General structure and domain organization of TdT. (A)
Linear arrangement of the different domains of C-TdT (blue),
compared with pol  .
The two motifs C and A and the two HhH consensus sequences are
indicated in red and green, respectively. The disordered part of
C-TdT in the crystal is in cyan and the N-terminal extension of
C-TdT compared with pol .
The two motifs C and A and the two HhH consensus sequences are
indicated in red and green, respectively. The disordered part of
C-TdT in the crystal is in cyan and the N-terminal extension of
C-TdT compared with pol  is
in magenta. The part of the molecule absent from the
crystallized construct is in white. (B) General architecture of
the TdT structure drawn with Molscript (Kraulis, 1991). The
three catalytic aspartate residues are shown in ball-and-stick
representation. The N- and C-termini are in dark blue, while the
two strictly conserved stretches of sequences implicated in the
binding of the incoming dNTP are in cyan and magenta. Loop1 is
shown in yellow. An intrinsic magnesium ion (grey) as well as
the two putative sodium ions bound to the HhH motifs are
represented in CPK mode (blue); only the Na^+ ion with ligands
in an octahedral geometry is labelled. is
in magenta. The part of the molecule absent from the
crystallized construct is in white. (B) General architecture of
the TdT structure drawn with Molscript (Kraulis, 1991). The
three catalytic aspartate residues are shown in ball-and-stick
representation. The N- and C-termini are in dark blue, while the
two strictly conserved stretches of sequences implicated in the
binding of the incoming dNTP are in cyan and magenta. Loop1 is
shown in yellow. An intrinsic magnesium ion (grey) as well as
the two putative sodium ions bound to the HhH motifs are
represented in CPK mode (blue); only the Na^+ ion with ligands
in an octahedral geometry is labelled.
|
 |
Figure 3.
Figure 3 C-TdT is in a closed form. Stereo view of the
superimposed C[  ]traces
of TdT (red) with the closed form of pol ]traces
of TdT (red) with the closed form of pol  (blue). (blue).
|
 |
|
|

|
| |
The above figures are
reprinted
from an Open Access publication published by Macmillan Publishers Ltd:
EMBO J
(2002,
21,
427-439)
copyright 2002.
|
|
| |
Figures were
selected
by an automated process.
|
|

|

|
|
 |
 |
|
 |
 |
 |
 |
 |
 |
 |
 |
 |
|
Literature references that cite this PDB file's key reference
|
|
 |
| |
PubMed id
|
 |
Reference
|
 |
|
|
|
 |
P.Xie
(2011).
A model for the dynamics of mammalian family X DNA polymerases.
|
| |
J Theor Biol,
277,
111-122.
|
 |
|
|
|
|
 |
S.K.Jarchow-Choy,
A.T.Krueger,
H.Liu,
J.Gao,
and
E.T.Kool
(2011).
Fluorescent xDNA nucleotides as efficient substrates for a template-independent polymerase.
|
| |
Nucleic Acids Res,
39,
1586-1594.
|
 |
|
|
|
|
 |
E.A.Motea,
and
A.J.Berdis
(2010).
Terminal deoxynucleotidyl transferase: the story of a misguided DNA polymerase.
|
| |
Biochim Biophys Acta,
1804,
1151-1166.
|
 |
|
|
|
|
 |
K.Bebenek,
M.Garcia-Diaz,
R.Z.Zhou,
L.F.Povirk,
and
T.A.Kunkel
(2010).
Loop 1 modulates the fidelity of DNA polymerase lambda.
|
| |
Nucleic Acids Res,
38,
5419-5431.
|
 |
|
PDB codes:
|
 |
|
|
|
|
|
 |
Y.Li,
and
T.Schlick
(2010).
Modeling DNA polymerase μ motions: subtle transitions before chemistry.
|
| |
Biophys J,
99,
3463-3472.
|
 |
|
|
|
|
 |
B.Gozalbo-López,
P.Andrade,
G.Terrados,
B.de Andrés,
N.Serrano,
I.Cortegano,
B.Palacios,
A.Bernad,
L.Blanco,
M.A.Marcos,
and
M.L.Gaspar
(2009).
A role for DNA polymerase mu in the emerging DJH rearrangements of the postgastrulation mouse embryo.
|
| |
Mol Cell Biol,
29,
1266-1275.
|
 |
|
|
|
|
 |
F.Romain,
I.Barbosa,
J.Gouge,
F.Rougeon,
and
M.Delarue
(2009).
Conferring a template-dependent polymerase activity to terminal deoxynucleotidyltransferase by mutations in the Loop1 region.
|
| |
Nucleic Acids Res,
37,
4642-4656.
|
 |
|
|
|
|
 |
M.Morar,
K.Bhullar,
D.W.Hughes,
M.Junop,
and
G.D.Wright
(2009).
Structure and mechanism of the lincosamide antibiotic adenylyltransferase LinB.
|
| |
Structure,
17,
1649-1659.
|
 |
|
PDB codes:
|
 |
|
|
|
|
|
 |
M.W.Voehler,
R.L.Eoff,
W.H.McDonald,
F.P.Guengerich,
and
M.P.Stone
(2009).
Modulation of the structure, catalytic activity, and fidelity of african Swine Fever virus DNA polymerase x by a reversible disulfide switch.
|
| |
J Biol Chem,
284,
18434-18444.
|
 |
|
|
|
|
 |
P.Andrade,
M.J.Martín,
R.Juárez,
F.López de Saro,
and
L.Blanco
(2009).
Limited terminal transferase in human DNA polymerase mu defines the required balance between accuracy and efficiency in NHEJ.
|
| |
Proc Natl Acad Sci U S A,
106,
16203-16208.
|
 |
|
|
|
|
 |
A.F.Moon,
M.Garcia-Diaz,
K.Bebenek,
B.J.Davis,
X.Zhong,
D.A.Ramsden,
T.A.Kunkel,
and
L.C.Pedersen
(2007).
Structural insight into the substrate specificity of DNA Polymerase mu.
|
| |
Nat Struct Mol Biol,
14,
45-53.
|
 |
|
PDB code:
|
 |
|
|
|
|
|
 |
A.F.Moon,
M.Garcia-Diaz,
V.K.Batra,
W.A.Beard,
K.Bebenek,
T.A.Kunkel,
S.H.Wilson,
and
L.C.Pedersen
(2007).
The X family portrait: structural insights into biological functions of X family polymerases.
|
| |
DNA Repair (Amst),
6,
1709-1725.
|
 |
|
|
|
|
 |
A.J.Berdis,
and
D.McCutcheon
(2007).
The use of non-natural nucleotides to probe template-independent DNA synthesis.
|
| |
Chembiochem,
8,
1399-1408.
|
 |
|
|
|
|
 |
G.C.Lin,
J.Jaeger,
and
J.B.Sweasy
(2007).
Loop II of DNA polymerase beta is important for polymerization activity and fidelity.
|
| |
Nucleic Acids Res,
35,
2924-2935.
|
 |
|
|
|
|
 |
G.Martin,
and
W.Keller
(2007).
RNA-specific ribonucleotidyl transferases.
|
| |
RNA,
13,
1834-1849.
|
 |
|
|
|
|
 |
J.Gu,
H.Lu,
B.Tippin,
N.Shimazaki,
M.F.Goodman,
and
M.R.Lieber
(2007).
XRCC4:DNA ligase IV can ligate incompatible DNA ends and can ligate across gaps.
|
| |
EMBO J,
26,
1010-1023.
|
 |
|
|
|
|
 |
J.M.Yoshizawa,
C.Li,
and
P.D.Gershon
(2007).
Saltatory forward movement of a poly(A) polymerase during poly(A) tail addition.
|
| |
J Biol Chem,
282,
19144-19151.
|
 |
|
|
|
|
 |
J.Stagno,
I.Aphasizheva,
A.Rosengarth,
H.Luecke,
and
R.Aphasizhev
(2007).
UTP-bound and Apo structures of a minimal RNA uridylyltransferase.
|
| |
J Mol Biol,
366,
882-899.
|
 |
|
PDB codes:
|
 |
|
|
|
|
|
 |
P.B.Balbo,
and
A.Bohm
(2007).
Mechanism of poly(A) polymerase: structure of the enzyme-MgATP-RNA ternary complex and kinetic analysis.
|
| |
Structure,
15,
1117-1131.
|
 |
|
PDB code:
|
 |
|
|
|
|
|
 |
S.Beetz,
D.Diekhoff,
and
L.A.Steiner
(2007).
Characterization of terminal deoxynucleotidyl transferase and polymerase mu in zebrafish.
|
| |
Immunogenetics,
59,
735-744.
|
 |
|
|
|
|
 |
T.Kubota,
S.Maezawa,
K.Koiwai,
T.Hayano,
and
O.Koiwai
(2007).
Identification of functional domains in TdIF1 and its inhibitory mechanism for TdT activity.
|
| |
Genes Cells,
12,
941-959.
|
 |
|
|
|
|
 |
B.Pardo,
E.Ma,
and
S.Marcand
(2006).
Mismatch tolerance by DNA polymerase Pol4 in the course of nonhomologous end joining in Saccharomyces cerevisiae.
|
| |
Genetics,
172,
2689-2694.
|
 |
|
|
|
|
 |
M.C.Foley,
K.Arora,
and
T.Schlick
(2006).
Sequential side-chain residue motions transform the binary into the ternary state of DNA polymerase lambda.
|
| |
Biophys J,
91,
3182-3195.
|
 |
|
|
|
|
 |
R.Juárez,
J.F.Ruiz,
S.A.Nick McElhinny,
D.Ramsden,
and
L.Blanco
(2006).
A specific loop in human DNA polymerase mu allows switching between creative and DNA-instructed synthesis.
|
| |
Nucleic Acids Res,
34,
4572-4582.
|
 |
|
|
|
|
 |
E.Crespan,
S.Zanoli,
A.Khandazhinskaya,
I.Shevelev,
M.Jasko,
L.Alexandrova,
M.Kukhanova,
G.Blanca,
G.Villani,
U.Hübscher,
S.Spadari,
and
G.Maga
(2005).
Incorporation of non-nucleoside triphosphate analogues opposite to an abasic site by human DNA polymerases beta and lambda.
|
| |
Nucleic Acids Res,
33,
4117-4127.
|
 |
|
|
|
|
 |
H.D.Cho,
C.L.Verlinde,
and
A.M.Weiner
(2005).
Archaeal CCA-adding enzymes: central role of a highly conserved beta-turn motif in RNA polymerization without translocation.
|
| |
J Biol Chem,
280,
9555-9566.
|
 |
|
|
|
|
 |
J.Deng,
N.L.Ernst,
S.Turley,
K.D.Stuart,
and
W.G.Hol
(2005).
Structural basis for UTP specificity of RNA editing TUTases from Trypanosoma brucei.
|
| |
EMBO J,
24,
4007-4017.
|
 |
|
PDB codes:
|
 |
|
|
|
|
|
 |
M.K.Kukhanova,
A.V.Ivanov,
and
M.V.Jasko
(2005).
Structural-functional relationships between terminal deoxynucleotidyltransferase and 5'-triphosphates of nucleoside analogs.
|
| |
Biochemistry (Mosc),
70,
890-896.
|
 |
|
|
|
|
 |
S.A.Nick McElhinny,
J.M.Havener,
M.Garcia-Diaz,
R.Juárez,
K.Bebenek,
B.L.Kee,
L.Blanco,
T.A.Kunkel,
and
D.A.Ramsden
(2005).
A gradient of template dependence defines distinct biological roles for family X polymerases in nonhomologous end joining.
|
| |
Mol Cell,
19,
357-366.
|
 |
|
|
|
|
 |
S.González-Barrera,
A.Sánchez,
J.F.Ruiz,
R.Juárez,
A.J.Picher,
G.Terrados,
P.Andrade,
and
L.Blanco
(2005).
Characterization of SpPol4, a unique X-family DNA polymerase in Schizosaccharomyces pombe.
|
| |
Nucleic Acids Res,
33,
4762-4774.
|
 |
|
|
|
|
 |
T.T.Paull
(2005).
Saving the ends for last: the role of pol mu in DNA end joining.
|
| |
Mol Cell,
19,
294-296.
|
 |
|
|
|
|
 |
A.Zhelkovsky,
S.Helmling,
A.Bohm,
and
C.Moore
(2004).
Mutations in the middle domain of yeast poly(A) polymerase affect interactions with RNA but not ATP.
|
| |
RNA,
10,
558-564.
|
 |
|
|
|
|
 |
B.Tippin,
S.Kobayashi,
J.G.Bertram,
and
M.F.Goodman
(2004).
To slip or skip, visualizing frameshift mutation dynamics for error-prone DNA polymerases.
|
| |
J Biol Chem,
279,
45360-45368.
|
 |
|
|
|
|
 |
K.Ramadan,
I.Shevelev,
and
U.Hübscher
(2004).
The DNA-polymerase-X family: controllers of DNA quality?
|
| |
Nat Rev Mol Cell Biol,
5,
1038-1043.
|
 |
|
|
|
|
 |
M.Garcia-Diaz,
K.Bebenek,
J.M.Krahn,
L.Blanco,
T.A.Kunkel,
and
L.C.Pedersen
(2004).
A structural solution for the DNA polymerase lambda-dependent repair of DNA gaps with minimal homology.
|
| |
Mol Cell,
13,
561-572.
|
 |
|
PDB code:
|
 |
|
|
|
|
|
 |
P.Schimmel,
and
X.L.Yang
(2004).
Two classes give lessons about CCA.
|
| |
Nat Struct Mol Biol,
11,
807-808.
|
 |
|
|
|
|
 |
S.A.Nick McElhinny,
and
D.A.Ramsden
(2004).
Sibling rivalry: competition between Pol X family members in V(D)J recombination and general double strand break repair.
|
| |
Immunol Rev,
200,
156-164.
|
 |
|
|
|
|
 |
C.Lehmann,
K.Lim,
V.R.Chalamasetty,
W.Krajewski,
E.Melamud,
A.Galkin,
A.Howard,
Z.Kelman,
P.T.Reddy,
A.G.Murzin,
and
O.Herzberg
(2003).
The HI0073/HI0074 protein pair from Haemophilus influenzae is a member of a new nucleotidyltransferase family: structure, sequence analyses, and solution studies.
|
| |
Proteins,
50,
249-260.
|
 |
|
PDB code:
|
 |
|
|
|
|
|
 |
I.Shevelev,
G.Blanca,
G.Villani,
K.Ramadan,
S.Spadari,
U.Hübscher,
and
G.Maga
(2003).
Mutagenesis of human DNA polymerase lambda: essential roles of Tyr505 and Phe506 for both DNA polymerase and terminal transferase activities.
|
| |
Nucleic Acids Res,
31,
6916-6925.
|
 |
|
|
|
|
 |
J.F.Ruiz,
R.Juárez,
M.García-Díaz,
G.Terrados,
A.J.Picher,
S.González-Barrera,
A.R.Fernández de Henestrosa,
and
L.Blanco
(2003).
Lack of sugar discrimination by human Pol mu requires a single glycine residue.
|
| |
Nucleic Acids Res,
31,
4441-4449.
|
 |
|
|
|
|
 |
K.Fujita,
N.Shimazaki,
Y.Ohta,
T.Kubota,
S.Ibe,
S.Toji,
K.Tamai,
S.Fujisaki,
T.Hayano,
and
O.Koiwai
(2003).
Terminal deoxynucleotidyltransferase forms a ternary complex with a novel chromatin remodeling protein with 82 kDa and core histone.
|
| |
Genes Cells,
8,
559-571.
|
 |
|
|
|
|
 |
N.Ito,
O.Nureki,
M.Shirouzu,
S.Yokoyama,
and
F.Hanaoka
(2003).
Crystal structure of the Pyrococcus horikoshii DNA primase-UTP complex: implications for the mechanism of primer synthesis.
|
| |
Genes Cells,
8,
913-923.
|
 |
|
PDB codes:
|
 |
|
|
|
|
|
 |
R.Hartmann,
J.Justesen,
S.N.Sarkar,
G.C.Sen,
and
V.C.Yee
(2003).
Crystal structure of the 2'-specific and double-stranded RNA-activated interferon-induced antiviral protein 2'-5'-oligoadenylate synthetase.
|
| |
Mol Cell,
12,
1173-1185.
|
 |
|
PDB code:
|
 |
|
|
|
|
|
 |
Y.Xiong,
F.Li,
J.Wang,
A.M.Weiner,
and
T.A.Steitz
(2003).
Crystal structures of an archaeal class I CCA-adding enzyme and its nucleotide complexes.
|
| |
Mol Cell,
12,
1165-1172.
|
 |
|
PDB codes:
|
 |
|
|
|
|
|
 |
F.Li,
Y.Xiong,
J.Wang,
H.D.Cho,
K.Tomita,
A.M.Weiner,
and
T.A.Steitz
(2002).
Crystal structures of the Bacillus stearothermophilus CCA-adding enzyme and its complexes with ATP or CTP.
|
| |
Cell,
111,
815-824.
|
 |
|
PDB codes:
|
 |
|
|
 |
 |
|
The most recent references are shown first.
Citation data come partly from CiteXplore and partly
from an automated harvesting procedure. Note that this is likely to be
only a partial list as not all journals are covered by
either method. However, we are continually building up the citation data
so more and more references will be included with time.
Where a reference describes a PDB structure, the PDB
codes are
shown on the right.
|
 |

