
|

|

|
|
 |
Contents |
 |
|
|

|

|
|
|
|
|
|
|
|
* Residue conservation analysis
|
|
|
 |
|
|
 |
 |
 |
 |
Enzyme class 1:
|
 |
Chains A, B:
E.C.2.7.7.-
- ?????
|
|
 |
 |
 |
 |
 |
Enzyme class 2:
|
 |
Chains A, B:
E.C.2.7.7.49
- RNA-directed Dna polymerase.
|
|
 |
 |
 |
 |
 |
Reaction:
|
 |
DNA(n) + a 2'-deoxyribonucleoside 5'-triphosphate = DNA(n+1) + diphosphate
|
 |
 |
 |
 |
 |
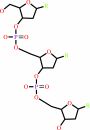 DNA(n)
DNA(n)
|
+
|
2'-deoxyribonucleoside 5'-triphosphate
|
=
|
DNA(n+1)
|
+
|
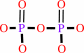 diphosphate
diphosphate
|
|
 |
 |
 |
 |
 |
 |
 |
 |
Enzyme class 3:
|
 |
Chains A, B:
E.C.2.7.7.7
- DNA-directed Dna polymerase.
|
|
 |
 |
 |
 |
 |
Reaction:
|
 |
DNA(n) + a 2'-deoxyribonucleoside 5'-triphosphate = DNA(n+1) + diphosphate
|
 |
 |
 |
 |
 |
 DNA(n)
DNA(n)
|
+
|
2'-deoxyribonucleoside 5'-triphosphate
|
=
|
DNA(n+1)
|
+
|
 diphosphate
diphosphate
|
|
 |
 |
 |
 |
 |
 |
 |
 |
Enzyme class 4:
|
 |
Chains A, B:
E.C.3.1.-.-
|
|
 |
 |
 |
 |
 |
Enzyme class 5:
|
 |
Chains A, B:
E.C.3.1.13.2
- exoribonuclease H.
|
|
 |
 |
 |
 |
 |
Reaction:
|
 |
Exonucleolytic cleavage to 5'-phosphomonoester oligonucleotides in both 5'- to 3'- and 3'- to 5'-directions.
|
 |
 |
 |
 |
 |
Enzyme class 6:
|
 |
Chains A, B:
E.C.3.1.26.13
- retroviral ribonuclease H.
|
|
 |
 |
 |
 |
 |
Enzyme class 7:
|
 |
Chains A, B:
E.C.3.4.23.16
- HIV-1 retropepsin.
|
|
 |
 |
 |
 |
 |
Reaction:
|
 |
Specific for a P1 residue that is hydrophobic, and P1' variable, but often Pro.
|
 |
 |
 |
 |
 |
 |
 |
|
Note, where more than one E.C. class is given (as above), each may
correspond to a different protein domain or, in the case of polyprotein
precursors, to a different mature protein.
|
|
 |
|
Molecule diagrams generated from .mol files obtained from the
KEGG ftp site
|
|
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |

|

|

|
| |
|
|
| |
|
DOI no:
|
J Biol Chem
275:14316-14320
(2000)
|
|
PubMed id:
|
|
|
|

|
| |
|
Binding of the second generation non-nucleoside inhibitor S-1153 to HIV-1 reverse transcriptase involves extensive main chain hydrogen bonding.
|
|
J.Ren,
C.Nichols,
L.E.Bird,
T.Fujiwara,
H.Sugimoto,
D.I.Stuart,
D.K.Stammers.
|
|
|

|
| |
ABSTRACT
|
|

|
| |

|
S-1153 (AG1549) is perhaps the most promising non-nucleoside inhibitor of HIV-1
reverse transcriptase currently under development as a potential anti-AIDS drug,
because it has a favorable profile of resilience to many drug resistance
mutations. We have determined the crystal structure of S-1153 in a complex with
HIV-1 reverse transcriptase. The complex possesses some novel features,
including an extensive network of hydrogen bonds involving the main chain of
residues 101, 103, and 236 of the p66 reverse transcriptase subunit. Such
interactions are unlikely to be disrupted by side chain mutations. The reverse
transcriptase/S-1153 complex suggests different ways in which resilience to
mutations in the non-nucleoside inhibitors of reverse transcriptase binding site
can be achieved.

|

|
|
|

|
| |
Selected figure(s)
|
|

|
| |
 |
 |

|
 |

|
 |
Figure 1.
Fig. 1. Simulated annealing omit electron density map
showing the bound S-1153 in the NNRTI pocket of HIV-1 RT. The
map is contoured at 3  . .
|
 |
Figure 3.
Fig. 3. Schematic diagram showing the intermolecular
interactions between S-1153 and the surrounding residues of
HIV-1 RT. The pink spheres represent water molecules in contact
with the inhibitor. Residues that contact the NNRTI with a
minimum interatomic distance of  3.6Å are
shown in green, whereas other residues lining the binding pocket
are shown in blue. The individual distances between the NNRTI
and the protein atoms are shown as dashed lines (distances less
than or equal to 3.3 Å are in pink; distances between 3.3
and 3.6 Å are in light blue; hydrogen bonds are shown
together with distances in black). 3.6Å are
shown in green, whereas other residues lining the binding pocket
are shown in blue. The individual distances between the NNRTI
and the protein atoms are shown as dashed lines (distances less
than or equal to 3.3 Å are in pink; distances between 3.3
and 3.6 Å are in light blue; hydrogen bonds are shown
together with distances in black).
|
 |
|
|

|
| |
The above figures are
reprinted
by permission from the ASBMB:
J Biol Chem
(2000,
275,
14316-14320)
copyright 2000.
|
|
| |
Figures were
selected
by an automated process.
|
|

|

|
|
 |
 |
|
 |
 |
 |
 |
 |
 |
 |
 |
 |
|
Literature references that cite this PDB file's key reference
|
|
 |
| |
PubMed id
|
 |
Reference
|
 |
|
|
|
 |
L.H.Jones,
G.Allan,
R.Corbau,
D.S.Middleton,
C.E.Mowbray,
S.D.Newman,
C.Phillips,
R.Webster,
and
M.Westby
(2011).
Comparison of the non-nucleoside reverse transcriptase inhibitor lersivirine with its pyrazole and imidazole isomers.
|
| |
Chem Biol Drug Des,
77,
393-397.
|
 |
|
|
|
|
 |
J.J.Tan,
X.J.Cong,
L.M.Hu,
C.X.Wang,
L.Jia,
and
X.J.Liang
(2010).
Therapeutic strategies underpinning the development of novel techniques for the treatment of HIV infection.
|
| |
Drug Discov Today,
15,
186-197.
|
 |
|
|
|
|
 |
K.Singh,
B.Marchand,
K.A.Kirby,
E.Michailidis,
and
S.G.Sarafianos
(2010).
Structural Aspects of Drug Resistance and Inhibition of HIV-1 Reverse Transcriptase.
|
| |
Viruses,
2,
606-638.
|
 |
|
|
|
|
 |
A.Ivetac,
and
J.A.McCammon
(2009).
Elucidating the inhibition mechanism of HIV-1 non-nucleoside reverse transcriptase inhibitors through multicopy molecular dynamics simulations.
|
| |
J Mol Biol,
388,
644-658.
|
 |
|
|
|
|
 |
Z.K.Sweeney,
J.J.Kennedy-Smith,
J.Wu,
N.Arora,
J.R.Billedeau,
J.P.Davidson,
J.Fretland,
J.Q.Hang,
G.M.Heilek,
S.F.Harris,
D.Hirschfeld,
P.Inbar,
H.Javanbakht,
J.A.Jernelius,
Q.Jin,
Y.Li,
W.Liang,
R.Roetz,
K.Sarma,
M.Smith,
D.Stefanidis,
G.Su,
J.M.Suh,
A.G.Villaseñor,
M.Welch,
F.J.Zhang,
and
K.Klumpp
(2009).
Diphenyl ether non-nucleoside reverse transcriptase inhibitors with excellent potency against resistant mutant viruses and promising pharmacokinetic properties.
|
| |
ChemMedChem,
4,
88-99.
|
 |
|
|
|
|
 |
D.T.Manallack
(2008).
The use of local surface properties for molecular superimposition.
|
| |
J Mol Model,
14,
797-805.
|
 |
|
|
|
|
 |
G.Barreiro,
C.R.Guimarães,
I.Tubert-Brohman,
T.M.Lyons,
J.Tirado-Rives,
and
W.L.Jorgensen
(2007).
Search for non-nucleoside inhibitors of HIV-1 reverse transcriptase using chemical similarity, molecular docking, and MM-GB/SA scoring.
|
| |
J Chem Inf Model,
47,
2416-2428.
|
 |
|
|
|
|
 |
A.Lavecchia,
R.Costi,
M.Artico,
G.Miele,
E.Novellino,
A.Bergamini,
E.Crespan,
G.Maga,
and
R.Di Santo
(2006).
Arylthiopyrrole (AThP) derivatives as non-nucleoside HIV-1 reverse transcriptase inhibitors: synthesis, structure-activity relationships, and docking studies (part 2).
|
| |
ChemMedChem,
1,
1379-1390.
|
 |
|
|
|
|
 |
C.Fortin,
V.Joly,
and
P.Yeni
(2006).
Emerging reverse transcriptase inhibitors for the treatment of HIV infection in adults.
|
| |
Expert Opin Emerg Drugs,
11,
217-230.
|
 |
|
|
|
|
 |
D.C.Meadows,
and
J.Gervay-Hague
(2006).
Current developments in HIV chemotherapy.
|
| |
ChemMedChem,
1,
16-29.
|
 |
|
|
|
|
 |
R.Di Santo,
R.Costi,
M.Artico,
G.Miele,
A.Lavecchia,
E.Novellino,
A.Bergamini,
R.Cancio,
and
G.Maga
(2006).
Arylthiopyrrole (AThP) derivatives as non-nucleoside HIV-1 reverse transcriptase inhibitors: synthesis, structure-activity relationships, and docking studies (part 1).
|
| |
ChemMedChem,
1,
1367-1378.
|
 |
|
|
|
|
 |
E.De Clercq
(2005).
Emerging anti-HIV drugs.
|
| |
Expert Opin Emerg Drugs,
10,
241-273.
|
 |
|
|
|
|
 |
J.L.Medina-Franco,
A.Golbraikh,
S.Oloff,
R.Castillo,
and
A.Tropsha
(2005).
Quantitative structure-activity relationship analysis of pyridinone HIV-1 reverse transcriptase inhibitors using the k nearest neighbor method and QSAR-based database mining.
|
| |
J Comput Aided Mol Des,
19,
229-242.
|
 |
|
|
|
|
 |
E.N.Peletskaya,
A.A.Kogon,
S.Tuske,
E.Arnold,
and
S.H.Hughes
(2004).
Nonnucleoside inhibitor binding affects the interactions of the fingers subdomain of human immunodeficiency virus type 1 reverse transcriptase with DNA.
|
| |
J Virol,
78,
3387-3397.
|
 |
|
PDB code:
|
 |
|
|
|
|
|
 |
F.Daeyaert,
M.de Jonge,
J.Heeres,
L.Koymans,
P.Lewi,
M.H.Vinkers,
and
P.A.Janssen
(2004).
A pharmacophore docking algorithm and its application to the cross-docking of 18 HIV-NNRTI's in their binding pockets.
|
| |
Proteins,
54,
526-533.
|
 |
|
|
|
|
 |
J.D.Pata,
W.G.Stirtan,
S.W.Goldstein,
and
T.A.Steitz
(2004).
Structure of HIV-1 reverse transcriptase bound to an inhibitor active against mutant reverse transcriptases resistant to other nonnucleoside inhibitors.
|
| |
Proc Natl Acad Sci U S A,
101,
10548-10553.
|
 |
|
PDB code:
|
 |
|
|
|
|
|
 |
N.Sluis-Cremer,
N.A.Temiz,
and
I.Bahar
(2004).
Conformational changes in HIV-1 reverse transcriptase induced by nonnucleoside reverse transcriptase inhibitor binding.
|
| |
Curr HIV Res,
2,
323-332.
|
 |
|
|
|
|
 |
S.J.Teague
(2003).
Implications of protein flexibility for drug discovery.
|
| |
Nat Rev Drug Discov,
2,
527-541.
|
 |
|
|
|
|
 |
E.De Clercq
(2002).
Strategies in the design of antiviral drugs.
|
| |
Nat Rev Drug Discov,
1,
13-25.
|
 |
|
|
|
|
 |
E.De Clercq
(2002).
New anti-HIV agents and targets.
|
| |
Med Res Rev,
22,
531-565.
|
 |
|
|
|
|
 |
J.Lindberg,
S.Sigurdsson,
S.Löwgren,
H.O.Andersson,
C.Sahlberg,
R.Noréen,
K.Fridborg,
H.Zhang,
and
T.Unge
(2002).
Structural basis for the inhibitory efficacy of efavirenz (DMP-266), MSC194 and PNU142721 towards the HIV-1 RT K103N mutant.
|
| |
Eur J Biochem,
269,
1670-1677.
|
 |
|
PDB codes:
|
 |
|
|
|
|
|
 |
L.Menéndez-Arias
(2002).
Targeting HIV: antiretroviral therapy and development of drug resistance.
|
| |
Trends Pharmacol Sci,
23,
381-388.
|
 |
|
|
|
|
 |
E.N.Peletskaya,
P.L.Boyer,
A.A.Kogon,
P.Clark,
H.Kroth,
J.M.Sayer,
D.M.Jerina,
and
S.H.Hughes
(2001).
Cross-linking of the fingers subdomain of human immunodeficiency virus type 1 reverse transcriptase to template-primer.
|
| |
J Virol,
75,
9435-9445.
|
 |
|
|
|
|
 |
J.Ren,
J.Milton,
K.L.Weaver,
S.A.Short,
D.I.Stuart,
and
D.K.Stammers
(2000).
Structural basis for the resilience of efavirenz (DMP-266) to drug resistance mutations in HIV-1 reverse transcriptase.
|
| |
Structure,
8,
1089-1094.
|
 |
|
PDB codes:
|
 |
|
|
 |
 |
|
The most recent references are shown first.
Citation data come partly from CiteXplore and partly
from an automated harvesting procedure. Note that this is likely to be
only a partial list as not all journals are covered by
either method. However, we are continually building up the citation data
so more and more references will be included with time.
Where a reference describes a PDB structure, the PDB
code is
shown on the right.
|
 |

