 |
PDBsum entry 1dmp

|
|
|

|
 |

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|
 |

|

|

|
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |

|

|

|

|

|

|

|

|

|
|
Aspartyl proteinase
|
PDB id
|
|

|

|
1dmp
|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|
|
 |
Contents |
 |
|
|

|

|
|
|
|
|
|
* Residue conservation analysis
|
|
|
|
 |
|
|
 |
 |
 |
 |
Enzyme class 1:
|
 |
E.C.2.7.7.-
- ?????
|
|
 |
 |
 |
 |
 |
Enzyme class 2:
|
 |
E.C.2.7.7.49
- RNA-directed Dna polymerase.
|
|
 |
 |
 |
 |
 |
Reaction:
|
 |
DNA(n) + a 2'-deoxyribonucleoside 5'-triphosphate = DNA(n+1) + diphosphate
|
 |
 |
 |
 |
 |
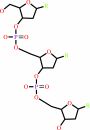 DNA(n)
DNA(n)
|
+
|
2'-deoxyribonucleoside 5'-triphosphate
|
=
|
DNA(n+1)
|
+
|
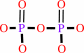 diphosphate
diphosphate
|
|
 |
 |
 |
 |
 |
 |
 |
 |
Enzyme class 3:
|
 |
E.C.2.7.7.7
- DNA-directed Dna polymerase.
|
|
 |
 |
 |
 |
 |
Reaction:
|
 |
DNA(n) + a 2'-deoxyribonucleoside 5'-triphosphate = DNA(n+1) + diphosphate
|
 |
 |
 |
 |
 |
 DNA(n)
DNA(n)
|
+
|
2'-deoxyribonucleoside 5'-triphosphate
|
=
|
DNA(n+1)
|
+
|
 diphosphate
diphosphate
|
|
 |
 |
 |
 |
 |
 |
 |
 |
Enzyme class 4:
|
 |
E.C.3.1.-.-
|
|
 |
 |
 |
 |
 |
Enzyme class 5:
|
 |
E.C.3.1.13.2
- exoribonuclease H.
|
|
 |
 |
 |
 |
 |
Reaction:
|
 |
Exonucleolytic cleavage to 5'-phosphomonoester oligonucleotides in both 5'- to 3'- and 3'- to 5'-directions.
|
 |
 |
 |
 |
 |
Enzyme class 6:
|
 |
E.C.3.1.26.13
- retroviral ribonuclease H.
|
|
 |
 |
 |
 |
 |
Enzyme class 7:
|
 |
E.C.3.4.23.16
- HIV-1 retropepsin.
|
|
 |
 |
 |
 |
 |
Reaction:
|
 |
Specific for a P1 residue that is hydrophobic, and P1' variable, but often Pro.
|
 |
 |
 |
 |
 |
 |
 |
|
Note, where more than one E.C. class is given (as above), each may
correspond to a different protein domain or, in the case of polyprotein
precursors, to a different mature protein.
|
|
 |
|
Molecule diagrams generated from .mol files obtained from the
KEGG ftp site
|
|
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |

|

|

|
| |
|
|
| |
|
|
Chem Biol
3:301-314
(1996)
|
|
PubMed id:
|
|
|
|

|
| |
|
Improved cyclic urea inhibitors of the HIV-1 protease: synthesis, potency, resistance profile, human pharmacokinetics and X-ray crystal structure of DMP 450.
|
|
C.N.Hodge,
P.E.Aldrich,
L.T.Bacheler,
C.H.Chang,
C.J.Eyermann,
S.Garber,
M.Grubb,
D.A.Jackson,
P.K.Jadhav,
B.Korant,
P.Y.Lam,
M.B.Maurin,
J.L.Meek,
M.J.Otto,
M.M.Rayner,
C.Reid,
T.R.Sharpe,
L.Shum,
D.L.Winslow,
S.Erickson-Viitanen.
|
|
|

|
| |
ABSTRACT
|
|

|
| |

|
BACKGROUND: Effective HIV protease inhibitors must combine potency towards
wild-type and mutant variants of HIV with oral bioavailability such that drug
levels in relevant tissues continuously exceed that required for inhibition of
virus replication. Computer-aided design led to the discovery of cyclic urea
inhibitors of the HIV protease. We set out to improve the physical properties
and oral bioavailability of these compounds. RESULTS: We have synthesized DMP
450 (bis-methanesulfonic acid salt), a water-soluble cyclic urea compound and a
potent inhibitor of HIV replication in cell culture that also inhibits variants
of HIV with single amino acid substitutions in the protease. DMP 450 is highly
selective for HIV protease, consistent with displacement of the
retrovirus-specific structural water molecule. Single doses of 10 mg kg-1 DMP
450 result in plasma levels in man in excess of that required to inhibit
wild-type and several mutant HIVs. A plasmid-based, in vivo assay model suggests
that maintenance of plasma levels of DMP 450 near the antiviral IC90 suppresses
HIV protease activity in the animal. We did identify mutants that are resistant
to DMP 450, however; multiple mutations within the protease gene caused a
significant reduction in the antiviral response. CONCLUSIONS: DMP 450 is a
significant advance within the cyclic urea class of HIV protease inhibitors due
to its exceptional oral bioavailability. The data presented here suggest that an
optimal cyclic urea will provide clinical benefit in treating AIDS if it
combines favorable pharmacokinetics with potent activity against not only single
mutants of HIV, but also multiply-mutant variants.

|

|
|
|

|

|
|
 |
 |
|
 |
 |
 |
 |
 |
 |
 |
 |
 |
|
Literature references that cite this PDB file's key reference
|
|
 |
| |
PubMed id
|
 |
Reference
|
 |
|
|
|
 |
J.Chen,
M.Yang,
G.Hu,
S.Shi,
C.Yi,
and
Q.Zhang
(2009).
Insights into the functional role of protonation states in the HIV-1 protease-BEA369 complex: molecular dynamics simulations and free energy calculations.
|
| |
J Mol Model,
15,
1245-1252.
|
 |
|
|
|
|
 |
A.Tripathi,
M.Fornabaio,
F.Spyrakis,
A.Mozzarelli,
P.Cozzini,
and
G.E.Kellogg
(2007).
Complexity in modeling and understanding protonation states: computational titration of HIV-1-protease-inhibitor complexes.
|
| |
Chem Biodivers,
4,
2564-2577.
|
 |
|
|
|
|
 |
Y.F.Wang,
Y.Tie,
P.I.Boross,
J.Tozser,
A.K.Ghosh,
R.W.Harrison,
and
I.T.Weber
(2007).
Potent new antiviral compound shows similar inhibition and structural interactions with drug resistant mutants and wild type HIV-1 protease.
|
| |
J Med Chem,
50,
4509-4515.
|
 |
|
PDB codes:
|
 |
|
|
|
|
|
 |
Z.Li,
and
T.Lazaridis
(2007).
Water at biomolecular binding interfaces.
|
| |
Phys Chem Chem Phys,
9,
573-581.
|
 |
|
|
|
|
 |
A.Y.Kovalevsky,
Y.Tie,
F.Liu,
P.I.Boross,
Y.F.Wang,
S.Leshchenko,
A.K.Ghosh,
R.W.Harrison,
and
I.T.Weber
(2006).
Effectiveness of nonpeptide clinical inhibitor TMC-114 on HIV-1 protease with highly drug resistant mutations D30N, I50V, and L90M.
|
| |
J Med Chem,
49,
1379-1387.
|
 |
|
PDB codes:
|
 |
|
|
|
|
|
 |
N.P.Todorov,
C.L.Buenemann,
and
I.L.Alberts
(2006).
De novo ligand design to an ensemble of protein structures.
|
| |
Proteins,
64,
43-59.
|
 |
|
|
|
|
 |
V.Frecer,
E.Burello,
and
S.Miertus
(2005).
Combinatorial design of nonsymmetrical cyclic urea inhibitors of aspartic protease of HIV-1.
|
| |
Bioorg Med Chem,
13,
5492-5501.
|
 |
|
|
|
|
 |
B.C.Logsdon,
J.F.Vickrey,
P.Martin,
G.Proteasa,
J.I.Koepke,
S.R.Terlecky,
Z.Wawrzak,
M.A.Winters,
T.C.Merigan,
and
L.C.Kovari
(2004).
Crystal structures of a multidrug-resistant human immunodeficiency virus type 1 protease reveal an expanded active-site cavity.
|
| |
J Virol,
78,
3123-3132.
|
 |
|
PDB codes:
|
 |
|
|
|
|
|
 |
J.H.Sun,
J.A.Lemm,
D.R.O'Boyle,
J.Racela,
R.Colonno,
and
M.Gao
(2003).
Specific inhibition of bovine viral diarrhea virus replicase.
|
| |
J Virol,
77,
6753-6760.
|
 |
|
|
|
|
 |
M.Kumar,
and
M.V.Hosur
(2003).
Adaptability and flexibility of HIV-1 protease.
|
| |
Eur J Biochem,
270,
1231-1239.
|
 |
|
|
|
|
 |
A.Wlodawer
(2002).
Rational approach to AIDS drug design through structural biology.
|
| |
Annu Rev Med,
53,
595-614.
|
 |
|
|
|
|
 |
E.De Clercq
(2002).
New anti-HIV agents and targets.
|
| |
Med Res Rev,
22,
531-565.
|
 |
|
|
|
|
 |
G.Moyle
(2002).
Overcoming obstacles to the success of protease inhibitors in highly active antiretroviral therapy regimens.
|
| |
AIDS Patient Care STDS,
16,
585-597.
|
 |
|
|
|
|
 |
C.A.Chen,
S.M.Sieburth,
A.Glekas,
G.W.Hewitt,
G.L.Trainor,
S.Erickson-Viitanen,
S.S.Garber,
B.Cordova,
S.Jeffry,
and
R.M.Klabe
(2001).
Drug design with a new transition state analog of the hydrated carbonyl: silicon-based inhibitors of the HIV protease.
|
| |
Chem Biol,
8,
1161-1166.
|
 |
|
|
|
|
 |
E.De Clercq
(2000).
Novel compounds in preclinical/early clinical development for the treatment of HIV infections.
|
| |
Rev Med Virol,
10,
255-277.
|
 |
|
|
|
|
 |
R.R.Speck,
C.Flexner,
C.J.Tian,
and
X.F.Yu
(2000).
Comparison of human immunodeficiency virus type 1 Pr55(Gag) and Pr160(Gag-pol) processing intermediates that accumulate in primary and transformed cells treated with peptidic and nonpeptidic protease inhibitors.
|
| |
Antimicrob Agents Chemother,
44,
1397-1403.
|
 |
|
|
|
|
 |
A.Hilgeroth
(1998).
[HIV-1 protease inhibitors in review]
|
| |
Pharm Unserer Zeit,
27,
22-25.
|
 |
|
|
|
|
 |
A.K.Patick,
and
K.E.Potts
(1998).
Protease inhibitors as antiviral agents.
|
| |
Clin Microbiol Rev,
11,
614-627.
|
 |
|
|
|
|
 |
A.Molla,
G.R.Granneman,
E.Sun,
and
D.J.Kempf
(1998).
Recent developments in HIV protease inhibitor therapy.
|
| |
Antiviral Res,
39,
1.
|
 |
|
|
|
|
 |
A.Wlodawer,
and
J.Vondrasek
(1998).
Inhibitors of HIV-1 protease: a major success of structure-assisted drug design.
|
| |
Annu Rev Biophys Biomol Struct,
27,
249-284.
|
 |
|
|
|
|
 |
J.D.Rodgers,
P.Y.Lam,
B.L.Johnson,
H.Wang,
R.Li,
Y.Ru,
S.S.Ko,
S.P.Seitz,
G.L.Trainor,
P.S.Anderson,
R.M.Klabe,
L.T.Bacheler,
B.Cordova,
S.Garber,
C.Reid,
M.R.Wright,
C.H.Chang,
and
S.Erickson-Viitanen
(1998).
Design and selection of DMP 850 and DMP 851: the next generation of cyclic urea HIV protease inhibitors.
|
| |
Chem Biol,
5,
597-608.
|
 |
|
|
|
|
 |
M.Patel,
L.T.Bacheler,
M.M.Rayner,
B.C.Cordova,
R.M.Klabe,
S.Erickson-Viitanen,
and
S.P.Seitz
(1998).
The synthesis and evaluation of cyclic ureas as HIV protease inhibitors: modifications of the P1/P1' residues.
|
| |
Bioorg Med Chem Lett,
8,
823-828.
|
 |
|
|
|
|
 |
P.J.Ala,
R.J.DeLoskey,
E.E.Huston,
P.K.Jadhav,
P.Y.Lam,
C.J.Eyermann,
C.N.Hodge,
M.C.Schadt,
F.A.Lewandowski,
P.C.Weber,
D.D.McCabe,
J.L.Duke,
and
C.H.Chang
(1998).
Molecular recognition of cyclic urea HIV-1 protease inhibitors.
|
| |
J Biol Chem,
273,
12325-12331.
|
 |
|
PDB code:
|
 |
|
|
|
|
|
 |
R.M.Klabe,
L.T.Bacheler,
P.J.Ala,
S.Erickson-Viitanen,
and
J.L.Meek
(1998).
Resistance to HIV protease inhibitors: a comparison of enzyme inhibition and antiviral potency.
|
| |
Biochemistry,
37,
8735-8742.
|
 |
|
|
|
|
 |
P.J.Ala,
E.E.Huston,
R.M.Klabe,
D.D.McCabe,
J.L.Duke,
C.J.Rizzo,
B.D.Korant,
R.J.DeLoskey,
P.Y.Lam,
C.N.Hodge,
and
C.H.Chang
(1997).
Molecular basis of HIV-1 protease drug resistance: structural analysis of mutant proteases complexed with cyclic urea inhibitors.
|
| |
Biochemistry,
36,
1573-1580.
|
 |
|
PDB codes:
|
 |
|
|
|
|
|
 |
U.Nillroth,
L.Vrang,
P.O.Markgren,
J.Hultén,
A.Hallberg,
and
U.H.Danielson
(1997).
Human immunodeficiency virus type 1 proteinase resistance to symmetric cyclic urea inhibitor analogs.
|
| |
Antimicrob Agents Chemother,
41,
2383-2388.
|
 |
|
 |
 |
|
The most recent references are shown first.
Citation data come partly from CiteXplore and partly
from an automated harvesting procedure. Note that this is likely to be
only a partial list as not all journals are covered by
either method. However, we are continually building up the citation data
so more and more references will be included with time.
Where a reference describes a PDB structure, the PDB
codes are
shown on the right.
|
 |

