Documentation
Residue/Cofactor Functions
On the first annotation page, the user simply types in the names of the amino acid residues involved in the reaction. All the amino acid residues drawn in the mechanism should be included in the list, irrespective of whether they are actively involved in that step or not as long as they are involved in some step of the reaction.
Amino acid residues are named using their three letter code and their number in the sequence is assigned from the PDB file. If amino acids from multiple chains are involved, the chains are differentiated with the chain identifier as from the PDB file, e.g. Glu56B, Asp185A, where Glu56 is from chain B, and Asp185 is from chain A. Once the initial annotation script is updated, the user must define which part of the amino acid is involved in the reaction and the functions that amino acid carries out.
Cofactors are treated in a very similar manner to amino acid residues, they are named using their HET group code and number in the sequence that is assigned from the PDB file. If the cofactors are from multiple chains, the chains are differentiated with the chain identifier from the PDB file, e.g. FAD444B, FMN445A, where FAD444 is from chain B, and FMN445 is from chain A. The further annotation requires the user to input the type of cofactor, and the functions it carries out.
Location of Residue Function
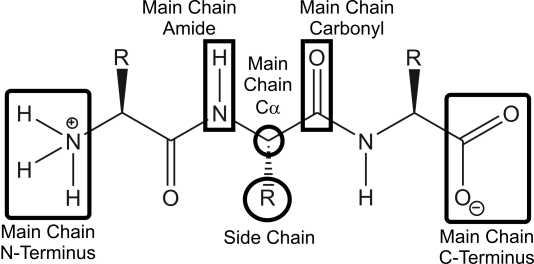 Legend:Location of amino acid residue function
Legend:Location of amino acid residue function-
The options for location of amino acid function are:
- Side Chain. The R' group of the amino acid.
- Post-translationally modification. Side chain of amino acid residues that have been post-translationally modified, and so may be considered "non-standard", e.g. a phosphrylated serine.
- Main Chain Amide. The N-H portion of the polypeptide backbone.
- Main Chain Carbonyl. The C=O portion of the polypeptide backbone.
- Main Chain Cα. The Cα portion of the polypeptide backbone.
- Main Chain C-Terminus. The C-terminal residue of the enzyme.
- Main Chain N-Terminus. The N-terminal residue of the enzyme.
Residue and Cofactor Function
-
There are three basic types of amino acid residue function:
- Interaction Types. This is the manner in which the amino acid residue or cofactor is interacting with other species in the reaction step. All amino acid residue annotations should have at least one interaction type.
- Spectator Functions. These are the functions that any amino acid residue or cofactor may perform. Spectator species are defined by including the onlooker term in their list of functions.
- Reactant Functions. These are the functions that only those amino acid residues or cofactors that are acting as reactants may perform.
It is also possible to assign a residue with an unknown function. This should only be assigned when the mechanism of the step is unclear, and this should be the only function assigned to any residue where the exact function and role is unknown.
Interaction Functions
The following are a list of the interaction types allowed in MACiE:
- Attractive Charge-Charge Interaction. Any charged residue that is involved in an attractive charge-charge interaction with another charged species.
- Covalently Attached. The amino acid residue (or cofactor) forms a covalent bond with another species involved in the reaction. The component may be permanently attached to the enzyme, or the interaction may be transient (i.e. formed and then cleaved during the course of the reaction mechanism).
- Hydrogen Bond Acceptor. Chemical species contains a nitrogen, oxygen, sulphur, or halogen atom with at least one available lone pair. This then forms a weak interaction with an entity containing a nitrogen, oxygen or sulphur atom which is covalently bound to at least one hydrogen atom.
- Hydrogen Bond Donor. Chemical species contains a nitrogen, oxygen or sulphur atom which is covalently bound to at least one hydrogen atom. This then forms a weak interaction with an entity which contains a nitrogen, oxygen, sulphur or halogen with at least one available lone pair.
- Hydrophobic Interaction. The tendency of hydrocarbons (or of lipophilic hydrocarbon-like groups in solutes) to form intermolecular aggregates in an aqueous medium, and analogous intramolecular interactions. The name arises from the attribution of the phenomenon to the apparent repulsion between water and hydrocarbons. However, the phenomenon ought to be attributed to the effect of the hydrocarbon-like groups on the water-water interaction. A pi-pi interaction is a special case of a hydrophobic interaction.
- Metal Ligand. The species is bound to a metal ion as a ligand of that ion. This function is only assigned when the amino acid or cofactor is catalytic, i.e. performing a simultaneous function. If a residue is only ligating a metal, it is not included in the annotation.
- Pi-Pi Interaction. pi-pi stacking interactions. Also known as pi-stacking.
- Polar Interaction. Any polar species which is involved in a polar-polar interaction with another polar species, including both attractive and repulsive forces. Interactions through hydrogen bonds are special cases of polar interactions.
- Polar/Non-Polar Interaction. Any polar or non-polar species which is involved in a non-covalent interaction with another non-polar or polar chemical species, respectively.
- Repulsive Charge-Charge Interaction. Any charged residue which is involved in a repulsive charge-charge interaction with another charged species.
- Van Der Waals Interaction. Any non-polar chemical species which is involved in a non-covalent interaction with another non-polar chemical species. This includes aromatic π-stacking and the stabilisation of non-polar radical species by the excess π-electrons of an aromatic species.
Spectator Functions
The following are a list of the spectator functions allowed in MACiE:
- Activator. Chemical species that increases the reactivity of another species. These amino acid residues either increase, or decrease, the pKa of another species which may be water, another residue, cofactor or substrate, thus activating it for the reaction to occur. It also includes amino acids which effect the electrostatics of other species. Where possible, one of the more specific activating terms should be used.
- Alter Redox Potential. The component alters (either increases or decreases) the redox potential of another component in the reaction.
- Decrease Redox Potential. The component decreases the redox potential of a redox active species in the reaction.
- Electrostatic Destabiliser. Chemical species that exerts a repulsive electrostatic interaction upon charges developing during the course of a reaction step.
- Electrostatic Interaction. i.e. They (de)stabilise the transition state, cofactors or reaction intermediates. This term also covers those amino acid residues which perturbate other catalytic species involved in the reaction (e.g. other residues, substrates/intermediates, cofactors or water) such that they are able to react more readily
- Electrostatic Stabiliser. Chemical species that exerts an attractive electrostatic interaction upon charges developing during the course of a reaction step.
- Enhance Reactivity. The component alters the reactivity of another component in the reaction.
- Ground State Destabiliser. The component functions by destabilising the energetics of the ground state complex.
- Increase Acidity. The species increased the ability of one of the reactive centres to donate a proton during the course of the reaction, this is an activating function.
- Increase Basicity. The species increased the ability of one of the reactive centres to accept a proton during the course of the reaction, this is an activating function.
- Increase Electrophilicity. The species increases the electrophilicity of one of the reactive centres by means of electrostatic interactions, this is an activating function.
- Increase Nucleophilicity. The species increases the nucleophilicity of one of the reactive centres by means of electrostatic interactions, this is an activating function.
- Increase Redox Potential. The component increases the redox potential of a redox active species in the reaction.
- Modifies Pka. The component alters the pKa of another component in the reaction.
- Promote Heterolysis. This species promotes the heterolytic cleavage of a bond in the reaction, this is an activating function.
- Promote Homolysis. This species promotes the homolytic cleavage of a bond in the reaction, this is an activating function.
- Radical Stabiliser. Chemical species which stabilises a free radical carrying species during the course of the reaction.
- Steric Hindrance. The component residue hinders access to the reacting chemical species by virtue of its bulk. This is a role often used in direct the stereochemistry of the reaction.
- Steric Locator. The component correctly locates the reacting species to induce the correct reaction.
- Steric Role. The chemical species perturbs the reactive species in a purely steric manner to induce reaction. This can be either by correctly locating reactive species, or perturbative by hindering certain conformations due to the bulk of the species. An example of a steric residue might be Tyr89 in MACiE entry M0062 (EC 5.4.99.2, methylmalonyl-CoA mutase) in which the tyrosine blocks formation of the incorrect stereoisomer.
- Transition State Destabiliser. A Component which is involved in destabilising the transition state formed during the course of a reaction
- Transition State Stabiliser. Chemical species that specifically stabilises the transition state of the reaction.
- Unknown. The residue is known to act as a spectator (a component which is not directly involved in the reaction, but is present and required) during the course of the reaction, but it's exact role is unknown.
Reactant Functions
The following are a list of the spectator functions allowed in MACiE:
- Covalent Catalysis. Any reaction where one component becomes covalently attached to a second, distinct component. This may be via a nucleophilic, electrophilic or radical mechanism. For the purposes of enzyme annotation, two amino acid residues within a single enzyme are considered to be distinct components.
- Electrofuge. A leaving group which does not carry away the bonding electrons.
- Electron Pair Acceptor. This is the chemical species which accepts two electrons during the course of a reaction. This may occur either as part of an electron relay chain in which two electrons are transferred, or as part of a covalent reaction, for example when a covalently attached lysine accepts a pair of electrons as part of the PLP reaction mechanism.
- Electron Pair Donor. This is the chemical species which donates two electrons during the course of a reaction. This may occur either as part of an electron relay chain in which two electrons are transferred, or as part of a covalent reaction, for example when a covalently attached lysine donates a pair of electrons as part of the PLP reaction mechanism.
- Electron Pair Relay. A component which both donates and accepts a pair of electrons during the course of a single reaction step
- Electron Pair Shuttle. A component which shuttles electrons in pairs during the course of the catalytic reaction.
- Electron Shuttle. A component which accepts and donates electrons (either singly or in pairs) during the course of the catalytic reaction (i.e. across all steps).
- Electron Tunneling Medium. The component is involved in the quantum tunnelling of an electron
- Electrophile. An electrophile is a chemical species that forms a bond to its reaction partner (the nucleophile) by accepting both bonding electrons from that reaction partner. This is synonymous to the term Lewis acid.
- Hydride Acceptor. A chemical species that accepts a hydride during the course of the reaction.
- Hydride Donor. A chemical species that donates a hydride during the course of the reaction.
- Hydride Relay. The reaction contains a hydride relay, i.e. a chemical species that both accepts and donates a H- during the course of the reaction.
- Hydride Shuttle. A component which accepts and donates a hydride anion (H<sup>-</sup>, a hydrogen atom with two electrons associated) during the course of the catalytic reaction (i.e. across all steps).
- Hydrogen Radical Acceptor. Chemical species which accepts a hydrogen atom during the course of the reaction.
- Hydrogen Radical Donor. Chemical species which donates a hydrogen atom during the course of the reaction.
- Hydrogen Radical Relay. Chemical species that both accepts and donates a hydrogen during the course of a reaction.
- Hydrogen Radical Shuttle. A component which accepts and donates a hydrogen radical (H<sup>.</sup>, a hydrogen atom with a single electron associated) during the course of the catalytic reaction (i.e. across all steps).
- Leaving Group (Radical). An atom or group (charged or uncharged) that becomes detached from an atom in what is considered to be the residual or main part of the substrate. This term is used for leaving groups which carry away only one of the bonding electrons.
- Nucleofuge. A nucleofuge is a leaving group which does carry away the bonding electrons.
- Nucleophile. A nucleophile is a chemical species that forms a bond to its reaction partner (the electrophile) by donating both bonding electrons. This is synonymous to the term Lewis Base.
- Proton Acceptor. Chemical species which accepts a proton from another chemical species involved in a reaction step. This term is synonymous with the terms general base and Brønsted base.
- Proton Donor. Chemical species which donates a proton to another residue, substrate, cofactor or other moiety involved in a reaction step. This term is synonymous with general acid and Brønsted acid.
- Proton Relay. The reaction contains a proton relay, i.e. a chemical species that acts as both Brønsted base and Brønsted acid during the course of the reaction. This term also applies to the residue that performs this role.
- Proton Shuttle (General Acid/Base). The component transfers protons around the protein during the course of the chemical reaction. This refers to general acid/base catalysis, and so incorporates any chemical species which accepts and donates a proton (H<sup>+</sup>, a hydrogen atom with no electrons associated) during the course of the catalytic reaction (i.e. across all steps).
- Radical Combinant. This is the chemical species which combines with another species in a radical reaction to form a covalent bonded species. This is typically used as cofactor annotation for flavins reacting with molecular oxygen to generate 4α-hydroperoxy species. This is also used as the function of residues in AdH2 reactions.
- Single Electron Acceptor. This is the chemical species which accepts one electron during the course of a reaction.
- Single Electron Donor. This is the chemical species which donates one electron during the course of a reaction.
- Single Electron Relay. A component which both donates and accepts a single electron during the course of a single reaction step
- Single Electron Shuttle. A component which shuttles single electrons during the course of the catalytic reaction.
- Unknown. The component is known to be critical in the reaction as a reactant (i.e. is a species which is consumed during the course of the observed chemical reaction), but its exact role is unknown.
A Note on the terminology of covalent catalysis in enzymes
In an addition reaction the product (which could also be called a Lewis adduct) is formed from the the attack of the nucleophile (Lewis base) on the electrophile (Lewis acid). When this adduct breaks down (e.g. in a substitution reaction) the leaving group that carries away the bonding electrons is called the nucleofuge and the other leaving group is called the electrofuge. This is described in the diagram below:
 Legend:The definition of the species involved in covalent catalysis.
Legend:The definition of the species involved in covalent catalysis.