 |
PDBsum entry 2wid

|

|

|
|
 |
Contents |
 |
|
|

|

|
|
|
|
|
|
|
|
|
|
* Residue conservation analysis
|
|
|

|
|
PDB id:
|
 |
|
 |
| Name: |
 |
Hydrolase
|
 |
|
Title:
|
 |
Nonaged form of human butyrylcholinesterase inhibited by tabun analogue ta1
|
|
Structure:
|
 |
Cholinesterase. Chain: a. Fragment: residues 29-557. Synonym: butyrylcholinesterase, acylcholine acylhydrolase, choline esterase ii, pseudocholinesterase. Engineered: yes. Mutation: yes. Other_details: s198 is phosphoramidylated
|
|
Source:
|
 |
Homo sapiens. Human. Organism_taxid: 9606. Expressed in: cricetulus griseus. Expression_system_taxid: 10029. Expression_system_cell_line: cho-k1.
|
|
Resolution:
|
 |
|
2.30Å
|
R-factor:
|
0.191
|
R-free:
|
0.255
|
|
|
Authors:
|
 |
E.Carletti,N.Aurbek,E.Gillon,M.Loiodice,Y.Nicolet,J.Fontecilla, P.Masson,H.Thiermann,F.Nachon,F.Worek
|
|
Key ref:
|
 |
E.Carletti
et al.
(2009).
Structure-activity analysis of aging and reactivation of human butyrylcholinesterase inhibited by analogues of tabun.
Biochem J,
421,
97.
PubMed id:
|
 |
|
Date:
|
 |
|
11-May-09
|
Release date:
|
19-May-09
|
|
|

|

|
|
PROCHECK
|

|

|
|
|
Headers
|
 |
|
|
References
|
|
|
|

|

|
|
P06276
(CHLE_HUMAN) -
Cholinesterase from Homo sapiens
|
|

|

|
Seq:
Struc:
|
 |
 |
 |
602 a.a.
527 a.a.*
|

|

|

|

|
|

|

|

|
 |
 |

|
|
Key: |
 |
 |
Secondary structure |
 |
 |
CATH domain |
 |
|
*
PDB and UniProt seqs differ
at 4 residue positions (black
crosses)
|
|

|
|
|
 |
|
|
 |
 |
 |
 |
Enzyme class:
|
 |
E.C.3.1.1.8
- cholinesterase.
|
|
 |
 |
 |
 |
 |
Reaction:
|
 |
an acylcholine + H2O = a carboxylate + choline + H+
|
 |
 |
 |
 |
 |
 acylcholine
acylcholine
|
+
|
H2O
|
=
|
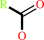 carboxylate
carboxylate
|
+
|
 choline
choline
|
+
|
H(+)
|
|
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
|
Molecule diagrams generated from .mol files obtained from the
KEGG ftp site
|
|
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |

|

|

|
| |
|
|
| |
|
|
Biochem J
421:97
(2009)
|
|
PubMed id:
|
|
|
|

|
| |
|
Structure-activity analysis of aging and reactivation of human butyrylcholinesterase inhibited by analogues of tabun.
|
|
E.Carletti,
N.Aurbek,
E.Gillon,
M.Loiodice,
Y.Nicolet,
J.C.Fontecilla-Camps,
P.Masson,
H.Thiermann,
F.Nachon,
F.Worek.
|
|
|

|
| |
ABSTRACT
|
|

|
| |

|
hBChE [human BChE (butyrylcholinesterase)] naturally scavenges OPs
(organophosphates). This bioscavenger is currently in Clinical Phase I for
pretreatment of OP intoxication. Phosphylated ChEs (cholinesterases) can undergo
a spontaneous time-dependent process called 'aging' during which the conjugate
is dealkylated, leading to creation of an enzyme that cannot be reactivated.
hBChE inhibited by phosphoramidates such as tabun displays a peculiar resistance
to oxime-mediated reactivation. We investigated the basis of oxime resistance of
phosphoramidyl-BChE conjugates by determining the kinetics of inhibition,
reactivation (obidoxime {1,1'-(oxybis-methylene) bis[4-(hydroxyimino) methyl]
pyridinium dichloride}, TMB-4
[1,3-trimethylene-bis(4-hydroxyiminomethylpyridinium) dibromide], HLö 7
{1-[[[4-(aminocarbonyl) pyridinio]methoxy]methyl]-2,4-bis-[(hydroxyimino)methyl]
pyridinium dimethanesulfonate)}, HI-6 {1-[[[4-(aminocarbonyl) pyridinio]
methoxy] methyl]-2-[(hydroxyimino)methyl]pyridinium dichloride monohydrate} and
aging, and the crystal structures of hBChE inhibited by different N-monoalkyl
and N,N-dialkyl tabun analogues. The refined structures of aged hBChE conjugates
show that aging proceeds through O-dealkylation of the P(R) enantiomer of
N,N-diethyl and N-propyl analogues, with subsequent formation of a salt bridge
preventing reactivation, similarly to a previous observation made on tabun-ChE
conjugates. Interestingly, the N-methyl analogue projects its amino group
towards the choline-binding pocket, so that aging proceeds through deamination.
This orientation results from a preference of hBChE's acyl-binding pocket for
larger than 2-atoms linear substituents. The correlation between the inhibitory
potency and the N-monoalkyl chain length is related to increasingly optimized
interactions with the acyl-binding pocket as shown by the X-ray structures.
These kinetics and X-ray data lead to a structure-activity relationship that
highlights steric and electronic effects of the amino substituent of
phosphoramidate. This study provides the structural basis to design new oximes
capable of reactivating phosphoramidyl-hBChE conjugates after intoxication,
notably when hBChE is used as pretreatment, or to design BChE-based catalytic
bioscavengers.

|

|
|
|

|

|
|
 |
 |
|
 |
 |
 |
 |
 |
 |
 |
 |
 |
|
Literature references that cite this PDB file's key reference
|
|
 |
| |
PubMed id
|
 |
Reference
|
 |
|
|
|
 |
F.Nachon,
E.Carletti,
M.Wandhammer,
Y.Nicolet,
L.M.Schopfer,
P.Masson,
and
O.Lockridge
(2011).
X-ray crystallographic snapshots of reaction intermediates in the G117H mutant of human butyrylcholinesterase, a nerve agent target engineered into a catalytic bioscavenger.
|
| |
Biochem J,
434,
73-82.
|
 |
|
PDB codes:
|
 |
|
|
|
|
|
 |
L.Wang,
D.Du,
D.Lu,
C.T.Lin,
J.N.Smith,
C.Timchalk,
F.Liu,
J.Wang,
and
Y.Lin
(2011).
Enzyme-linked immunosorbent assay for detection of organophosphorylated butyrylcholinesterase: a biomarker of exposure to organophosphate agents.
|
| |
Anal Chim Acta,
693,
1-6.
|
 |
|
|
|
|
 |
P.Masson,
and
O.Lockridge
(2010).
Butyrylcholinesterase for protection from organophosphorus poisons: catalytic complexities and hysteretic behavior.
|
| |
Arch Biochem Biophys,
494,
107-120.
|
 |
|
 |
 |
|
The most recent references are shown first.
Citation data come partly from CiteXplore and partly
from an automated harvesting procedure. Note that this is likely to be
only a partial list as not all journals are covered by
either method. However, we are continually building up the citation data
so more and more references will be included with time.
Where a reference describes a PDB structure, the PDB
codes are
shown on the right.
|
 |

