 |
PDBsum entry 1n7d

|
|
|

|
 |

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|
 |

|

|

|
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |

|

|

|

|

|

|

|

|

|
|
Lipid transport
|
PDB id
|
|

|

|
1n7d
|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|
|
 |
Contents |
 |
|
|

|

|
|
|
|
|
|
|
|
* Residue conservation analysis
|
|
|
|
|
References listed in PDB file
|
 |
|
Key reference
|
 |
|
Title
|
 |
Structure of the ldl receptor extracellular domain at endosomal ph.
|
 |
|
Authors
|
 |
G.Rudenko,
L.Henry,
K.Henderson,
K.Ichtchenko,
M.S.Brown,
J.L.Goldstein,
J.Deisenhofer.
|
 |
|
Ref.
|
 |
Science, 2002,
298,
2353-2358.
[DOI no: ]
|
 |
|
PubMed id
|
 |
|
 |
 |
|
Abstract
|
 |
|
The low-density lipoprotein receptor mediates cholesterol homeostasis through
endocytosis of lipoproteins. It discharges its ligand in the endosome at pH <
6. In the crystal structure at pH = 5.3, the ligand-binding domain (modules R2
to R7) folds back as an arc over the epidermal growth factor precursor homology
domain (the modules A, B, beta propeller, and C). The modules R4 and R5, which
are critical for lipoprotein binding, associate with the beta propeller via
their calcium-binding loop. We propose a mechanism for lipoprotein release in
the endosome whereby the beta propeller functions as an alternate substrate for
the ligand-binding domain, binding in a calcium-dependent way and promoting
lipoprotein release.
|
 |
 |
 |

|
 |

|
 |
Figure 2.
Fig. 2. Two monomers stabilized by tungsten clusters; the
ligand-binding domains have been labeled R2 to R7 and R2* to
R7*, respectively. The linkers between R4 and R5 and R4* and R5*
meet at a crystallographic twofold axis. At a distance below, a
cluster (center) sits on the twofold as well.
|
 |
Figure 3.
Fig. 3. Interface formed between R4, R5, and  propeller.
Residues discussed in the text are shown and labeled except for
Ser191, which was omitted for clarity. R4, magenta trace; R5,
red trace; propeller.
Residues discussed in the text are shown and labeled except for
Ser191, which was omitted for clarity. R4, magenta trace; R5,
red trace;  propeller,
cyan trace; and calcium ions, maroon spheres. propeller,
cyan trace; and calcium ions, maroon spheres.
|
 |
|
 |
 |
|
The above figures are
reprinted
by permission from the AAAs:
Science
(2002,
298,
2353-2358)
copyright 2002.
|
 |
|
Added reference #1*
|
 |
|
Title
|
 |
'MAD'ly phasing the extracellular domain of the LDL receptor: a medium-sized protein, large tungsten clusters and multiple non-isomorphous crystals.
|
 |
|
Authors
|
 |
G.Rudenko,
L.Henry,
C.Vonrhein,
G.Bricogne,
J.Deisenhofer.
|
 |
|
Ref.
|
 |
Acta Crystallogr D Biol Crystallogr, 2003,
59,
1978-1986.
[DOI no: ]
|
 |
|
PubMed id
|
 |
|
 |
 |
|
|
 |
 |
 |

|
 |

|
 |
Figure 1.
Figure 1 Modular organization of the extracellular region of
LDL-R. The `ligand-binding domain' and the `EGF precursor
homology domain' are indicated. The cysteine-rich repeats R1-R7
are separated by short linkers of approximately four residues,
with the exception of R4 and R5 which are separated by a
12-residue linker. The EGF-like repeats A, B and C flank the
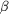 -propeller
domain. -propeller
domain.
|
 |
Figure 5.
Figure 5 Improvement of the electron-density map before (a) and
after (b) sharpening the data, as discussed in the text.
Examples are shown for residues Trp144, Glu219 and Asp299
(contoured at 1  )
and the 12-tungstophosphate cluster (contoured at 3.5 )
and the 12-tungstophosphate cluster (contoured at 3.5  ). ).
|
 |
|
 |
 |
|
The above figures are
reproduced from the cited reference
with permission from the IUCr
|
 |
 |
|
*Note, "added" references are those not in the PDB file but
which have either been obtained from the journal or suggested by the
author(s).
|
 |
|

|
|
|
 |

