 |
PDBsum entry 3fvq

|

|

|
|
 |
Contents |
 |
|
|

|

|
|
|
|
|
|
|
|
|
|
* Residue conservation analysis
|
|
|
|
 |
|
|
 |
 |
 |
 |
Enzyme class:
|
 |
E.C.7.2.2.7
- ABC-type Fe(3+) transporter.
|
|
 |
 |
 |
 |
 |
Reaction:
|
 |
Fe3+(out) + ATP + H2O = Fe3+(in) + ADP + phosphate + H+
|
 |
 |
 |
 |
 |
Fe(3+)(out)
Bound ligand (Het Group name = )
corresponds exactly
|
+
|
 ATP
ATP
|
+
|
H2O
|
=
|
Fe(3+)(in)
|
+
|
 ADP
ADP
|
+
|
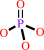 phosphate
phosphate
|
+
|
H(+)
|
|
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
|
Molecule diagrams generated from .mol files obtained from the
KEGG ftp site
|
|
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |

|

|

|
| |
|
|
| |
|
|
Structure
17:1213-1222
(2009)
|
|
PubMed id:
|
|
|
|

|
| |
|
Insights into how nucleotide-binding domains power ABC transport.
|
|
S.Newstead,
P.W.Fowler,
P.Bilton,
E.P.Carpenter,
P.J.Sadler,
D.J.Campopiano,
M.S.Sansom,
S.Iwata.
|
|
|

|
| |
ABSTRACT
|
|

|
| |

|
The mechanism by which nucleotide-binding domains (NBDs) of ABC transporters
power the transport of substrates across cell membranes is currently unclear.
Here we report the crystal structure of an NBD, FbpC, from the Neisseria
gonorrhoeae ferric iron uptake transporter with an unusual and substantial
domain swap in the C-terminal regulatory domain. This entanglement suggests that
FbpC is unable to open to the same extent as the homologous protein MalK. Using
molecular dynamics we demonstrate that this is not the case: both NBDs open
rapidly once ATP is removed. We conclude from this result that the closed
structures of FbpC and MalK have higher free energies than their respective open
states. This result has important implications for our understanding of the
mechanism of power generation in ABC transporters, because the unwinding of this
free energy ensures that the opening of these two NBDs is also powered.

|

|
|
|

|

|
|
 |
 |
|
 |
 |
 |
 |
 |
 |
 |
 |
 |
|
Literature references that cite this PDB file's key reference
|
|
 |
| |
PubMed id
|
 |
Reference
|
 |
|
|
|
 |
V.M.Korkhov,
S.A.Mireku,
and
K.P.Locher
(2012).
Structure of AMP-PNP-bound vitamin B12 transporter BtuCD-F.
|
| |
Nature,
490,
367-372.
|
 |
|
PDB code:
|
 |
|
|
 |
 |
|
The most recent references are shown first.
Citation data come partly from CiteXplore and partly
from an automated harvesting procedure. Note that this is likely to be
only a partial list as not all journals are covered by
either method. However, we are continually building up the citation data
so more and more references will be included with time.
Where a reference describes a PDB structure, the PDB
code is
shown on the right.
|
 |

