 |
PDBsum entry 2pjh

|
|
|

|
 |

|

|

|

|

|

|

|

|

|

|

|

|

|

|
 |

|

|

|
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |

|

|

|

|

|

|

|

|

|
|
Transport protein
|
PDB id
|
|

|

|
2pjh
|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|
|
 |
Contents |
 |
|
|

|

|
|
|
|
* Residue conservation analysis
|
|
|

|
|
PDB id:
|
 |
|
 |
| Name: |
 |
Transport protein
|
 |
|
Title:
|
 |
Strctural model of the p97 n domain- npl4 ubd complex
|
|
Structure:
|
 |
Nuclear protein localization protein 4 homolog. Chain: a. Fragment: n domain (residues 1-80). Synonym: protein npl4. Engineered: yes. Transitional endoplasmic reticulum atpase. Chain: b. Fragment: ubd (residues 21-213). Synonym: ter atpase, 15s mg2+, - atpase p97 subunit, valosin-
|
|
Source:
|
 |
Mus musculus. House mouse. Organism_taxid: 10090. Gene: p97. Expressed in: escherichia coli bl21. Expression_system_taxid: 511693. Gene: npl4. Expressed in: escherichia coli. Expression_system_taxid: 562
|
|
NMR struc:
|
 |
1 models
|
 |
|
Authors:
|
 |
R.Isaacson,V.E.Pye,S.Simpson,H.H.Meyer,X.Zhang,P.Freemont
|
Key ref:

|
 |
R.L.Isaacson
et al.
(2007).
Detailed structural insights into the p97-Npl4-Ufd1 interface.
J Biol Chem,
282,
21361-21369.
PubMed id:
DOI:

|
 |
|
Date:
|
 |
|
16-Apr-07
|
Release date:
|
08-May-07
|
|
|

|

|
|
PROCHECK
|

|

|
|
|
Headers
|
 |
|
|
References
|
|
|
|
|
 |
|
|
 |
 |
 |
 |
Enzyme class 1:
|
 |
Chain A:
E.C.?
|
|
 |
 |
 |
 |
 |
Enzyme class 2:
|
 |
Chain B:
E.C.3.6.4.6
- vesicle-fusing ATPase.
|
|
 |
 |
 |
 |
 |
Reaction:
|
 |
ATP + H2O = ADP + phosphate + H+
|
 |
 |
 |
 |
 |
 ATP
ATP
|
+
|
H2O
|
=
|
 ADP
ADP
|
+
|
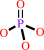 phosphate
phosphate
|
+
|
H(+)
|
|
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
|
Note, where more than one E.C. class is given (as above), each may
correspond to a different protein domain or, in the case of polyprotein
precursors, to a different mature protein.
|
|
 |
|
Molecule diagrams generated from .mol files obtained from the
KEGG ftp site
|
|
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |

|

|

|
| |
|
|
| |
|
DOI no:
|
J Biol Chem
282:21361-21369
(2007)
|
|
PubMed id:
|
|
|
|

|
| |
|
Detailed structural insights into the p97-Npl4-Ufd1 interface.
|
|
R.L.Isaacson,
V.E.Pye,
P.Simpson,
H.H.Meyer,
X.Zhang,
P.S.Freemont,
S.Matthews.
|
|
|

|
| |
ABSTRACT
|
|

|
| |

|
The AAA ATPase, p97, achieves its versatility through binding to a wide range of
cofactor proteins that adapt it to different cellular functions. The heterodimer
UN (comprising Ufd1 and Npl4) is an adaptor complex that recruits p97 for
numerous tasks, many of which involve the ubiquitin pathway. Insights into the
structural specificity of p97 for its UN adaptor are currently negligible. Here,
we present the solution structure of the Npl4 "ubiquitin-like" domain
(UBD), which adopts a beta-grasp fold with a 3(10) helical insert. Moreover we
performed a chemical shift perturbation analysis of its binding surface with the
p97 N domain. We assigned the backbone amides of the p97 N domain and probed
both its reciprocal binding surface with Npl4 UBD and its interaction with the
p97-binding region of Ufd1. NMR data recorded on a 400-kDa full-length
UN-hexamer p97 complex reveals an identical mode of interaction. We calculated a
structural model for the p97 N-Npl4 UBD complex, and a comparison with the
p97-p47 adaptor complex reveals subtle differences in p97 adaptor recognition
and specificity.

|

|
|
|

|
| |
Selected figure(s)
|
|

|
| |
 |
 |

|
 |

|
 |
Figure 5.
FIGURE 5. HADDOCK-derived model for the Npl4 UBD-p97 N
complex. A, C[  ]traces representing
the super-position of the 10 refined Npl4 UBD-p97 N complex
structures using the HADDOCK approach. B, ribbon representation
of the Npl4 UBD (pink)-p97 N (blue) complex. C, ribbon
representation of the p47 UBX (gold)-p97 N (blue) complex. The
coordinates for the p47 UBX-p97 N complex are taken from PDB
accession number 1S3S (20). D, close-up view of key interacting
side chains shown in stick representation. E, close-up view of
residues in Npl4 UBD, which, when mutated by Bruderer et al.
(15), abolished binding to p97 N domain. ]traces representing
the super-position of the 10 refined Npl4 UBD-p97 N complex
structures using the HADDOCK approach. B, ribbon representation
of the Npl4 UBD (pink)-p97 N (blue) complex. C, ribbon
representation of the p47 UBX (gold)-p97 N (blue) complex. The
coordinates for the p47 UBX-p97 N complex are taken from PDB
accession number 1S3S (20). D, close-up view of key interacting
side chains shown in stick representation. E, close-up view of
residues in Npl4 UBD, which, when mutated by Bruderer et al.
(15), abolished binding to p97 N domain.
|
 |
Figure 6.
FIGURE 6. Localization of the p97 N interaction motif
within Ufd1. A, ^1H-^13C HSQC spectra of the specifically
labeled Ufd1 (215-241) peptide (DHSGYAGEVGFRAFSGSGNRLDGKKKG,
^13C/^15N labels in bold and underlined) within increasing
amounts of p97 N. B, sequence alignment of the Ufd1 peptide with
p47 homologues illustrating the conserved nature of this motif.
|
 |
|
|

|
| |
The above figures are
reprinted
by permission from the ASBMB:
J Biol Chem
(2007,
282,
21361-21369)
copyright 2007.
|
|
| |
Figures were
selected
by an automated process.
|
|

|

|
|
 |
 |
|
 |
 |
 |
 |
 |
 |
 |
 |
 |
|
Literature references that cite this PDB file's key reference
|
|
 |
| |
PubMed id
|
 |
Reference
|
 |
|
|
|
 |
E.Chapman,
A.N.Fry,
and
M.Kang
(2011).
The complexities of p97 function in health and disease.
|
| |
Mol Biosyst,
7,
700-710.
|
 |
|
|
|
|
 |
C.Zhao,
E.A.Matveeva,
Q.Ren,
and
S.W.Whiteheart
(2010).
Dissecting the N-ethylmaleimide-sensitive factor: required elements of the N and D1 domains.
|
| |
J Biol Chem,
285,
761-772.
|
 |
|
|
|
|
 |
D.S.Haines
(2010).
p97-containing complexes in proliferation control and cancer: emerging culprits or guilt by association?
|
| |
Genes Cancer,
1,
753-763.
|
 |
|
|
|
|
 |
G.Zhao,
G.Li,
H.Schindelin,
and
W.J.Lennarz
(2009).
An Armadillo motif in Ufd3 interacts with Cdc48 and is involved in ubiquitin homeostasis and protein degradation.
|
| |
Proc Natl Acad Sci U S A,
106,
16197-16202.
|
 |
|
PDB code:
|
 |
|
|
|
|
|
 |
L.Madsen,
M.Seeger,
C.A.Semple,
and
R.Hartmann-Petersen
(2009).
New ATPase regulators--p97 goes to the PUB.
|
| |
Int J Biochem Cell Biol,
41,
2380-2388.
|
 |
|
|
|
|
 |
L.Madsen,
K.M.Andersen,
S.Prag,
T.Moos,
C.A.Semple,
M.Seeger,
and
R.Hartmann-Petersen
(2008).
Ubxd1 is a novel co-factor of the human p97 ATPase.
|
| |
Int J Biochem Cell Biol,
40,
2927-2942.
|
 |
|
|
|
|
 |
T.R.Heallen,
H.P.Adams,
T.Furuta,
K.J.Verbrugghe,
and
J.M.Schumacher
(2008).
An Afg2/Spaf-related Cdc48-like AAA ATPase regulates the stability and activity of the C. elegans Aurora B kinase AIR-2.
|
| |
Dev Cell,
15,
603-616.
|
 |
|
|
|
|
 |
R.L.Isaacson,
P.J.Simpson,
M.Liu,
E.Cota,
X.Zhang,
P.Freemont,
and
S.Matthews
(2007).
A new labeling method for methyl transverse relaxation-optimized spectroscopy NMR spectra of alanine residues.
|
| |
J Am Chem Soc,
129,
15428-15429.
|
 |
|
 |
 |
|
The most recent references are shown first.
Citation data come partly from CiteXplore and partly
from an automated harvesting procedure. Note that this is likely to be
only a partial list as not all journals are covered by
either method. However, we are continually building up the citation data
so more and more references will be included with time.
Where a reference describes a PDB structure, the PDB
code is
shown on the right.
|
 |
|

