 |
PDBsum entry 2f6f

|

|

|
|
 |
Contents |
 |
|
|

|

|
|
|
|
|
|
|
|
* Residue conservation analysis
|
|
|

|
|
PDB id:
|
 |
|
 |
| Name: |
 |
Hydrolase
|
 |
|
Title:
|
 |
The structure of the s295f mutant of human ptp1b
|
|
Structure:
|
 |
Tyrosine-protein phosphatase, non-receptor type 1. Chain: a. Fragment: catalytic domain. Synonym: protein-tyrosine phosphatase 1b, ptp-1b. Engineered: yes. Mutation: yes
|
|
Source:
|
 |
Homo sapiens. Human. Organism_taxid: 9606. Gene: ptpn1, ptp1b. Expressed in: escherichia coli bl21(de3). Expression_system_taxid: 469008.
|
|
Resolution:
|
 |
|
2.00Å
|
R-factor:
|
0.186
|
R-free:
|
0.203
|
|
|
Authors:
|
 |
J.Montalibet,K.Skorey,D.Mckay,G.Scapin,E.Asante-Appiah,B.P.Kennedy
|
Key ref:

|
 |
J.Montalibet
et al.
(2006).
Residues distant from the active site influence protein-tyrosine phosphatase 1B inhibitor binding.
J Biol Chem,
281,
5258-5266.
PubMed id:
DOI:

|
 |
|
Date:
|
 |
|
29-Nov-05
|
Release date:
|
06-Dec-05
|
|
|

|

|
|
PROCHECK
|

|

|
|
|
Headers
|
 |
|
|
References
|
|
|
|

|

|
|
P18031
(PTN1_HUMAN) -
Tyrosine-protein phosphatase non-receptor type 1 from Homo sapiens
|
|

|

|
Seq:
Struc:
|
 |
 |
 |
435 a.a.
302 a.a.*
|

|

|

|

|
|

|

|

|
 |
 |

|
|
Key: |
 |
PfamA domain |
 |
 |
 |
Secondary structure |
 |
 |
CATH domain |
 |
|
*
PDB and UniProt seqs differ
at 1 residue position (black
cross)
|
|

|
|
|
 |
|
|
 |
 |
 |
 |
Enzyme class:
|
 |
E.C.3.1.3.48
- protein-tyrosine-phosphatase.
|
|
 |
 |
 |
 |
 |
Reaction:
|
 |
O-phospho-L-tyrosyl-[protein] + H2O = L-tyrosyl-[protein] + phosphate
|
 |
 |
 |
 |
 |
O-phospho-L-tyrosyl-[protein]
|
+
|
H2O
|
=
|
L-tyrosyl-[protein]
|
+
|
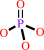 phosphate
phosphate
|
|
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
|
Molecule diagrams generated from .mol files obtained from the
KEGG ftp site
|
|
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |

|

|

|
| |
|
|
| |
|
DOI no:
|
J Biol Chem
281:5258-5266
(2006)
|
|
PubMed id:
|
|
|
|

|
| |
|
Residues distant from the active site influence protein-tyrosine phosphatase 1B inhibitor binding.
|
|
J.Montalibet,
K.Skorey,
D.McKay,
G.Scapin,
E.Asante-Appiah,
B.P.Kennedy.
|
|
|

|
| |
ABSTRACT
|
|

|
| |

|
Regions of protein-tyrosine phosphatase (PTP) 1B that are distant from the
active site yet affect inhibitor binding were identified by a novel library
screen. This screen was based on the observation that expression of v-Src in
yeast leads to lethality, which can be rescued by the coexpression of PTP1B.
However, this rescue is lost when yeast are grown in the presence of PTP1B
inhibitors. To identify regions of PTP1B (amino acids 1-400, catalytic domain
plus 80-amino acid C-terminal tail) that can affect the binding of the
difluoromethyl phosphonate (DFMP) inhibitor 7-bromo-6-difluoromethylphosphonate
3-naphthalenenitrile, a library coexpressing PTP1B mutants and v-Src was
generated, and the ability of yeast to grow in the presence of the inhibitor was
evaluated. PTP1B inhibitor-resistant mutations were found to concentrate on
helix alpha7 and its surrounding region, but not in the active site. No
resistant amino acid substitutions were found to occur in the C-terminal tail,
suggesting that this region has little effect on active-site inhibitor binding.
An in-depth characterization of a resistant substitution localizing to region
alpha7 (S295F) revealed that this change minimally affected enzyme catalytic
activity, but significantly reduced the potency of a panel of structurally
diverse DFMP PTP1B inhibitors. This loss of inhibitor potency was found to be
due to the difluoro moiety of these inhibitors because only the difluoro
inhibitors were shifted. For example, the inhibitor potency of a monofluorinated
or non-fluorinated analog of one of these DFMP inhibitors was only minimally
affected. Using this type of library screen, which can scan the nearly
full-length PTP1B sequence (catalytic domain and C-terminal tail) for effects on
inhibitor binding, we have been able to identify novel regions of PTP1B that
specifically affect the binding of DFMP inhibitors.

|

|
|
|

|
| |
Selected figure(s)
|
|

|
| |
 |
 |

|
 |

|
 |
Figure 1.
FIGURE 1. Modeling of Inhibitor 1 into the PTP1B active
site. The structure of Inhibitor 1 is shown in the inset. The
active-site nucleophile Cys^215 in the PTP loop (pink) is shown
along with Phe^182 in the WPD loop (green). Inhibitor 1 sits in
the active site with the phosphonate (red) interacting with the
PTP loop, and the difluoro group is within van der Waals
distance of the phenyl side chain of Phe^182.
|
 |
Figure 5.
FIGURE 5. Interactions of helix  7 with the PTP1B core.
In the wild-type structure (Protein Data Bank code 1SUG; shown
in cyan), an extensive network of hydrogen bonds connects helix 7 with the PTP1B core.
In the wild-type structure (Protein Data Bank code 1SUG; shown
in cyan), an extensive network of hydrogen bonds connects helix
 7
to helix 7
to helix  3 and the loop between
strands 3 and the loop between
strands  9 and 9 and  10. In
the S295F mutant (shown in yellow), helix 10. In
the S295F mutant (shown in yellow), helix  7 is repositioned and
partially disordered, with consequent disruption of this
extensive network of interactions. Only the hydrogen bond
between the side chains of Tyr^152 and Asn^193 is retained. 7 is repositioned and
partially disordered, with consequent disruption of this
extensive network of interactions. Only the hydrogen bond
between the side chains of Tyr^152 and Asn^193 is retained.
|
 |
|
|

|
| |
The above figures are
reprinted
by permission from the ASBMB:
J Biol Chem
(2006,
281,
5258-5266)
copyright 2006.
|
|
| |
Figures were
selected
by an automated process.
|
|

|

|
|
 |
 |
|
 |
 |
 |
 |
 |
 |
 |
 |
 |
|
Literature references that cite this PDB file's key reference
|
|
 |
| |
PubMed id
|
 |
Reference
|
 |
|
|
|
 |
S.G.Julien,
N.Dubé,
S.Hardy,
and
M.L.Tremblay
(2011).
Inside the human cancer tyrosine phosphatome.
|
| |
Nat Rev Cancer,
11,
35-49.
|
 |
|
|
|
|
 |
B.Townshend,
I.Aubry,
R.C.Marcellus,
K.Gehring,
and
M.L.Tremblay
(2010).
An RNA aptamer that selectively inhibits the enzymatic activity of protein tyrosine phosphatase 1B in vitro.
|
| |
Chembiochem,
11,
1583-1593.
|
 |
|
|
|
|
 |
K.Bharatham,
N.Bharatham,
Y.J.Kwon,
and
K.W.Lee
(2008).
Molecular dynamics simulation study of PTP1B with allosteric inhibitor and its application in receptor based pharmacophore modeling.
|
| |
J Comput Aided Mol Des,
22,
925-933.
|
 |
|
|
|
|
 |
L.Tabernero,
A.R.Aricescu,
E.Y.Jones,
and
S.E.Szedlacsek
(2008).
Protein tyrosine phosphatases: structure-function relationships.
|
| |
FEBS J,
275,
867-882.
|
 |
|
|
|
|
 |
S.G.Julien,
N.Dubé,
M.Read,
J.Penney,
M.Paquet,
Y.Han,
B.P.Kennedy,
W.J.Muller,
and
M.L.Tremblay
(2007).
Protein tyrosine phosphatase 1B deficiency or inhibition delays ErbB2-induced mammary tumorigenesis and protects from lung metastasis.
|
| |
Nat Genet,
39,
338-346.
|
 |
|
 |
 |
|
The most recent references are shown first.
Citation data come partly from CiteXplore and partly
from an automated harvesting procedure. Note that this is likely to be
only a partial list as not all journals are covered by
either method. However, we are continually building up the citation data
so more and more references will be included with time.
|
 |

