 |
PDBsum entry 1pxh

|

|

|
|
 |
Contents |
 |
|
|

|

|
|
|
|
|
|
|
|
|
|
* Residue conservation analysis
|
|
|

|
|
PDB id:
|
 |
|
 |
| Name: |
 |
Hydrolase
|
 |
|
Title:
|
 |
Crystal structure of protein tyrosine phosphatase 1b with potent and selective bidentate inhibitor compound 2
|
|
Structure:
|
 |
Protein-tyrosine phosphatase, non-receptor type 1. Chain: a. Fragment: residues 1-321. Synonym: protein-tyrosine phosphatase 1b, ptp-1b. Engineered: yes
|
|
Source:
|
 |
Homo sapiens. Human. Organism_taxid: 9606. Gene: ptp1b. Expressed in: escherichia coli bl21(de3). Expression_system_taxid: 469008.
|
|
Resolution:
|
 |
|
2.15Å
|
R-factor:
|
0.200
|
R-free:
|
0.207
|
|
|
Authors:
|
 |
J.P.Sun,A.Fedorov,S.Y.Lee,X.L.Guo,K.Shen,D.S.Lawrence,S.C.Almo, Z.Y.Zhang
|
Key ref:

|
 |
J.P.Sun
et al.
(2003).
Crystal structure of PTP1B complexed with a potent and selective bidentate inhibitor.
J Biol Chem,
278,
12406-12414.
PubMed id:
DOI:

|
 |
|
Date:
|
 |
|
04-Jul-03
|
Release date:
|
12-Aug-03
|
|
|
Supersedes:
|

|
|
|

|

|
|
PROCHECK
|

|

|
|
|
Headers
|
 |
|
|
References
|
|
|
|

|

|
|
P18031
(PTN1_HUMAN) -
Tyrosine-protein phosphatase non-receptor type 1 from Homo sapiens
|
|

|

|
Seq:
Struc:
|
 |
 |
 |
435 a.a.
297 a.a.
|

|

|

|

|
|

|

|

|
 |
 |

|
|
Key: |
 |
PfamA domain |
 |
 |
 |
Secondary structure |
 |
 |
CATH domain |
 |
|

|
|
|
 |
|
|
 |
 |
 |
 |
Enzyme class:
|
 |
E.C.3.1.3.48
- protein-tyrosine-phosphatase.
|
|
 |
 |
 |
 |
 |
Reaction:
|
 |
O-phospho-L-tyrosyl-[protein] + H2O = L-tyrosyl-[protein] + phosphate
|
 |
 |
 |
 |
 |
O-phospho-L-tyrosyl-[protein]
|
+
|
H2O
|
=
|
L-tyrosyl-[protein]
|
+
|
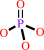 phosphate
phosphate
|
|
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
|
Molecule diagrams generated from .mol files obtained from the
KEGG ftp site
|
|
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |

|

|

|
| |
|
|
| |
|
DOI no:
|
J Biol Chem
278:12406-12414
(2003)
|
|
PubMed id:
|
|
|
|

|
| |
|
Crystal structure of PTP1B complexed with a potent and selective bidentate inhibitor.
|
|
J.P.Sun,
A.A.Fedorov,
S.Y.Lee,
X.L.Guo,
K.Shen,
D.S.Lawrence,
S.C.Almo,
Z.Y.Zhang.
|
|
|

|
| |
ABSTRACT
|
|

|
| |

|
Protein-tyrosine phosphatase 1B (PTP1B) has been implicated as an important
regulator in several signaling pathways including those initiated by insulin and
leptin. Potent and specific PTP1B inhibitors could serve as useful tools in
elucidating the physiological functions of PTP1B and may constitute valuable
therapeutics in the treatment of several human diseases. We have determined the
crystal structure of PTP1B in complex with compound 2, the most potent and
selective PTP1B inhibitor reported to date. The structure at 2.15-A resolution
reveals that compound 2 simultaneously binds to the active site and a unique
proximal noncatalytic site formed by Lys-41, Arg-47, and Asp-48. The structural
data are further corroborated by results from kinetic analyses of the
interactions of PTP1B and its site-directed mutants with compound 2 and several
of its variants. Although many of the residues important for interactions
between PTP1B and compound 2 are not unique to PTP1B, the combinations of all
contact residues differ between PTP isozymes, which provide a structural basis
for potent and selective PTP1B inhibition. Our data further suggest that potent,
yet highly selective, PTP1B inhibitory agents can be acquired by targeting the
area defined by residues Lys-41, Arg-47, and Asp-48, in addition to the
previously identified second aryl phosphate-binding pocket.

|

|
|
|

|
| |
Selected figure(s)
|
|

|
| |
 |
 |

|
 |

|
 |
Figure 4.
Fig. 4. Interactions between 2 and PTP1B. a, H-bonding
(blue dotted lines) and polar interactions (red dotted lines);
b, hydrophobic interactions (orange dotted lines), and; c,
interactions between the two fluorine atoms in F[2]Pmp and
PTP1B. The cut-off distance used is 3.2 Å for hydrogen
bonds, 4.0 Å for electrostatic or polar interactions, and
4.5 Å for hydrophobic interactions.
|
 |
Figure 5.
Fig. 5. Interactions of Arg-47 and Lys-41 with the EGFR
peptide (a), the PTP1B consensus peptide (b), compound 2 (c),
and compound 4 (d). Hydrophobic interactions are indicated by
orange dotted lines, H-bonds by blue dotted lines, and polar
interactions by red dotted lines.
|
 |
|
|

|
| |
The above figures are
reprinted
by permission from the ASBMB:
J Biol Chem
(2003,
278,
12406-12414)
copyright 2003.
|
|
| |
Figures were
selected
by an automated process.
|
|

|

|
|
 |
 |
|
 |
 |
 |
 |
 |
 |
 |
 |
 |
|
Literature references that cite this PDB file's key reference
|
|
 |
| |
PubMed id
|
 |
Reference
|
 |
|
|
|
 |
Y.S.Choong,
T.S.Lim,
A.L.Chew,
I.Aziah,
and
A.Ismail
(2011).
Structural and functional studies of a 50 kDa antigenic protein from Salmonella enterica serovar Typhi.
|
| |
J Mol Graph Model,
29,
834-842.
|
 |
|
|
|
|
 |
K.A.Rawls,
C.Grundner,
and
J.A.Ellman
(2010).
Design and synthesis of nonpeptidic, small molecule inhibitors for the Mycobacterium tuberculosis protein tyrosine phosphatase PtpB.
|
| |
Org Biomol Chem,
8,
4066-4070.
|
 |
|
|
|
|
 |
R.Huang,
I.Martinez-Ferrando,
and
P.A.Cole
(2010).
Enhanced interrogation: emerging strategies for cell signaling inhibition.
|
| |
Nat Struct Mol Biol,
17,
646-649.
|
 |
|
|
|
|
 |
T.Scior,
J.A.Guevara-García,
F.J.Melendez,
H.H.Abdallah,
Q.T.Do,
and
P.Bernard
(2010).
Chimeric design, synthesis, and biological assays of a new nonpeptide insulin-mimetic vanadium compound to inhibit protein tyrosine phosphatase 1B.
|
| |
Drug Des Devel Ther,
4,
231-242.
|
 |
|
|
|
|
 |
X.Zhang,
Y.He,
S.Liu,
Z.Yu,
Z.X.Jiang,
Z.Yang,
Y.Dong,
S.C.Nabinger,
L.Wu,
A.M.Gunawan,
L.Wang,
R.J.Chan,
and
Z.Y.Zhang
(2010).
Salicylic acid based small molecule inhibitor for the oncogenic Src homology-2 domain containing protein tyrosine phosphatase-2 (SHP2).
|
| |
J Med Chem,
53,
2482-2493.
|
 |
|
PDB codes:
|
 |
|
|
|
|
|
 |
R.Maccari,
R.Ottanà,
R.Ciurleo,
P.Paoli,
G.Manao,
G.Camici,
C.Laggner,
and
T.Langer
(2009).
Structure-based optimization of benzoic acids as inhibitors of protein tyrosine phosphatase 1B and low molecular weight protein tyrosine phosphatase.
|
| |
ChemMedChem,
4,
957-962.
|
 |
|
|
|
|
 |
T.Scior,
H.G.Mack,
J.A.García,
and
W.Koch
(2009).
Antidiabetic Bis-Maltolato-OxoVanadium(IV): Conversion of inactive trans- to bioactive cis-BMOV for possible binding to target PTP-1B.
|
| |
Drug Des Devel Ther,
2,
221-231.
|
 |
|
|
|
|
 |
K.Bharatham,
N.Bharatham,
Y.J.Kwon,
and
K.W.Lee
(2008).
Molecular dynamics simulation study of PTP1B with allosteric inhibitor and its application in receptor based pharmacophore modeling.
|
| |
J Comput Aided Mol Des,
22,
925-933.
|
 |
|
|
|
|
 |
K.Hellmuth,
S.Grosskopf,
C.T.Lum,
M.Würtele,
N.Röder,
J.P.von Kries,
M.Rosario,
J.Rademann,
and
W.Birchmeier
(2008).
Specific inhibitors of the protein tyrosine phosphatase Shp2 identified by high-throughput docking.
|
| |
Proc Natl Acad Sci U S A,
105,
7275-7280.
|
 |
|
|
|
|
 |
S.Liu,
L.F.Zeng,
L.Wu,
X.Yu,
T.Xue,
A.M.Gunawan,
Y.Q.Long,
and
Z.Y.Zhang
(2008).
Targeting inactive enzyme conformation: aryl diketoacid derivatives as a new class of PTP1B inhibitors.
|
| |
J Am Chem Soc,
130,
17075-17084.
|
 |
|
PDB codes:
|
 |
|
|
|
|
|
 |
Z.X.Jiang,
and
Z.Y.Zhang
(2008).
Targeting PTPs with small molecule inhibitors in cancer treatment.
|
| |
Cancer Metastasis Rev,
27,
263-272.
|
 |
|
|
|
|
 |
A.A.Puhl,
R.J.Gruninger,
R.Greiner,
T.W.Janzen,
S.C.Mosimann,
and
L.B.Selinger
(2007).
Kinetic and structural analysis of a bacterial protein tyrosine phosphatase-like myo-inositol polyphosphatase.
|
| |
Protein Sci,
16,
1368-1378.
|
 |
|
PDB codes:
|
 |
|
|
|
|
|
 |
C.D.Morrison,
C.L.White,
Z.Wang,
S.Y.Lee,
D.S.Lawrence,
W.T.Cefalu,
Z.Y.Zhang,
and
T.W.Gettys
(2007).
Increased hypothalamic protein tyrosine phosphatase 1B contributes to leptin resistance with age.
|
| |
Endocrinology,
148,
433-440.
|
 |
|
|
|
|
 |
J.Xie,
and
C.T.Seto
(2007).
A two stage click-based library of protein tyrosine phosphatase inhibitors.
|
| |
Bioorg Med Chem,
15,
458-473.
|
 |
|
|
|
|
 |
M.Stuible,
L.Zhao,
I.Aubry,
D.Schmidt-Arras,
F.D.Böhmer,
C.J.Li,
and
M.L.Tremblay
(2007).
Cellular inhibition of protein tyrosine phosphatase 1B by uncharged thioxothiazolidinone derivatives.
|
| |
Chembiochem,
8,
179-186.
|
 |
|
|
|
|
 |
S.C.Almo,
J.B.Bonanno,
J.M.Sauder,
S.Emtage,
T.P.Dilorenzo,
V.Malashkevich,
S.R.Wasserman,
S.Swaminathan,
S.Eswaramoorthy,
R.Agarwal,
D.Kumaran,
M.Madegowda,
S.Ragumani,
Y.Patskovsky,
J.Alvarado,
U.A.Ramagopal,
J.Faber-Barata,
M.R.Chance,
A.Sali,
A.Fiser,
Z.Y.Zhang,
D.S.Lawrence,
and
S.K.Burley
(2007).
Structural genomics of protein phosphatases.
|
| |
J Struct Funct Genomics,
8,
121-140.
|
 |
|
PDB codes:
|
 |
|
|
|
|
|
 |
X.Yu,
J.P.Sun,
Y.He,
X.Guo,
S.Liu,
B.Zhou,
A.Hudmon,
and
Z.Y.Zhang
(2007).
Structure, inhibitor, and regulatory mechanism of Lyp, a lymphoid-specific tyrosine phosphatase implicated in autoimmune diseases.
|
| |
Proc Natl Acad Sci U S A,
104,
19767-19772.
|
 |
|
|
|
|
 |
G.X.Liu,
J.Z.Tan,
C.Y.Niu,
J.H.Shen,
X.M.Luo,
X.Shen,
K.X.Chen,
and
H.L.Jiang
(2006).
Molecular dynamics simulations of interaction between protein-tyrosine phosphatase 1B and a bidentate inhibitor.
|
| |
Acta Pharmacol Sin,
27,
100-110.
|
 |
|
|
|
|
 |
A.K.Pedersen,
G.H.Peters G,
K.B.Møller,
L.F.Iversen,
and
J.S.Kastrup
(2004).
Water-molecule network and active-site flexibility of apo protein tyrosine phosphatase 1B.
|
| |
Acta Crystallogr D Biol Crystallogr,
60,
1527-1534.
|
 |
|
PDB code:
|
 |
|
|
|
|
|
 |
S.D.Taylor,
and
B.Hill
(2004).
Recent advances in protein tyrosine phosphatase 1B inhibitors.
|
| |
Expert Opin Investig Drugs,
13,
199-214.
|
 |
|
 |
 |
|
The most recent references are shown first.
Citation data come partly from CiteXplore and partly
from an automated harvesting procedure. Note that this is likely to be
only a partial list as not all journals are covered by
either method. However, we are continually building up the citation data
so more and more references will be included with time.
Where a reference describes a PDB structure, the PDB
codes are
shown on the right.
|
 |

