 |
PDBsum entry 1dv2

|

|

|
|
 |
Contents |
 |
|
|

|

|
|
|
|
|
|
|
|
* Residue conservation analysis
|
|
|
|
 |
|
|
 |
 |
 |
 |
Enzyme class:
|
 |
E.C.6.3.4.14
- biotin carboxylase.
|
|
 |
 |
 |
 |
 |
Reaction:
|
 |
N6-biotinyl-L-lysyl-[protein] + hydrogencarbonate + ATP = N6- carboxybiotinyl-L-lysyl-[protein] + ADP + phosphate + H+
|
 |
 |
 |
 |
 |
N(6)-biotinyl-L-lysyl-[protein]
Bound ligand (Het Group name = )
corresponds exactly
|
+
|
 hydrogencarbonate
hydrogencarbonate
|
+
|
 ATP
ATP
|
=
|
N(6)- carboxybiotinyl-L-lysyl-[protein]
|
+
|
 ADP
ADP
|
+
|
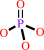 phosphate
phosphate
|
+
|
H(+)
|
|
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
|
Molecule diagrams generated from .mol files obtained from the
KEGG ftp site
|
|
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |

|

|

|
| |
|
|
| |
|
DOI no:
|
J Biol Chem
275:16183-16190
(2000)
|
|
PubMed id:
|
|
|
|

|
| |
|
Movement of the biotin carboxylase B-domain as a result of ATP binding.
|
|
J.B.Thoden,
C.Z.Blanchard,
H.M.Holden,
G.L.Waldrop.
|
|
|

|
| |
ABSTRACT
|
|

|
| |

|
Acetyl-CoA carboxylase catalyzes the first committed step in fatty acid
synthesis. In Escherichia coli, the enzyme is composed of three distinct protein
components: biotin carboxylase, biotin carboxyl carrier protein, and
carboxyltransferase. The biotin carboxylase component has served for many years
as a paradigm for mechanistic studies devoted toward understanding more
complicated biotin-dependent carboxylases. The three-dimensional x-ray structure
of an unliganded form of E. coli biotin carboxylase was originally solved in
1994 to 2.4-A resolution. This study revealed the architecture of the enzyme and
demonstrated that the protein belongs to the ATP-grasp superfamily. Here we
describe the three-dimensional structure of the E. coli biotin carboxylase
complexed with ATP and determined to 2.5-A resolution. The major conformational
change that occurs upon nucleotide binding is a rotation of approximately 45(o)
of one domain relative to the other domains thereby closing off the active site
pocket. Key residues involved in binding the nucleotide to the protein include
Lys-116, His-236, and Glu-201. The backbone amide groups of Gly-165 and Gly-166
participate in hydrogen bonding interactions with the phosphoryl oxygens of the
nucleotide. A comparison of this closed form of biotin carboxylase with
carbamoyl-phosphate synthetase is presented.

|

|
|
|

|
| |
Selected figure(s)
|
|

|
| |
 |
 |

|
 |

|
 |
Figure 2.
Fig. 2. Mode of binding of the inorganic phosphate ion to
biotin carboxylase. Shown in a is a representative portion of
the electron density map near the phosphate. The map, contoured
at 1  , was
calculated with coefficients of the form (2F[o] , was
calculated with coefficients of the form (2F[o]  F[c]),
where F[o] was the native structure factor amplitude and F[c]
was the calculated structure factor amplitude. The hydrogen
bonding pattern around the phosphate ion is indicated by the
dashed lines in b. Water molecules are depicted as red spheres. F[c]),
where F[o] was the native structure factor amplitude and F[c]
was the calculated structure factor amplitude. The hydrogen
bonding pattern around the phosphate ion is indicated by the
dashed lines in b. Water molecules are depicted as red spheres.
|
 |
Figure 5.
Fig. 5. Superposition of the  -carbon
trace of native biotin carboxylase onto that of the E288K
protein/ATP complex. The unliganded form of biotin carboxylase
is shown in red bonds, while the E288K protein is displayed in
black bonds. The ATP is depicted in a ball-and-stick
representation. -carbon
trace of native biotin carboxylase onto that of the E288K
protein/ATP complex. The unliganded form of biotin carboxylase
is shown in red bonds, while the E288K protein is displayed in
black bonds. The ATP is depicted in a ball-and-stick
representation.
|
 |
|
|

|
| |
The above figures are
reprinted
by permission from the ASBMB:
J Biol Chem
(2000,
275,
16183-16190)
copyright 2000.
|
|
| |
Figures were
selected
by an automated process.
|
|

|

|
|
 |
 |
|
 |
 |
 |
 |
 |
 |
 |
 |
 |
|
Literature references that cite this PDB file's key reference
|
|
 |
| |
PubMed id
|
 |
Reference
|
 |
|
|
|
 |
B.R.Novak,
D.Moldovan,
G.L.Waldrop,
and
M.S.de Queiroz
(2011).
Behavior of the ATP grasp domain of biotin carboxylase monomers and dimers studied using molecular dynamics simulations.
|
| |
Proteins,
79,
622-632.
|
 |
|
|
|
|
 |
C.S.Huang,
K.Sadre-Bazzaz,
Y.Shen,
B.Deng,
Z.H.Zhou,
and
L.Tong
(2010).
Crystal structure of the alpha(6)beta(6) holoenzyme of propionyl-coenzyme A carboxylase.
|
| |
Nature,
466,
1001-1005.
|
 |
|
PDB code:
|
 |
|
|
|
|
|
 |
J.O.Wrabl,
and
V.J.Hilser
(2010).
Investigating homology between proteins using energetic profiles.
|
| |
PLoS Comput Biol,
6,
e1000722.
|
 |
|
|
|
|
 |
P.K.Fyfe,
M.S.Alphey,
and
W.N.Hunter
(2010).
Structure of Trypanosoma brucei glutathione synthetase: domain and loop alterations in the catalytic cycle of a highly conserved enzyme.
|
| |
Mol Biochem Parasitol,
170,
93-99.
|
 |
|
PDB code:
|
 |
|
|
|
|
|
 |
C.Y.Chou,
L.P.Yu,
and
L.Tong
(2009).
Crystal structure of biotin carboxylase in complex with substrates and implications for its catalytic mechanism.
|
| |
J Biol Chem,
284,
11690-11697.
|
 |
|
PDB codes:
|
 |
|
|
|
|
|
 |
J.Matsui,
J.Nagano,
D.Miyoshi,
K.Tamaki,
and
N.Sugimoto
(2009).
An approach to peptide-based ATP receptors by a combination of random selection, rational design, and molecular imprinting.
|
| |
Biosens Bioelectron,
25,
563-567.
|
 |
|
|
|
|
 |
I.Mochalkin,
J.R.Miller,
A.Evdokimov,
S.Lightle,
C.Yan,
C.K.Stover,
and
G.L.Waldrop
(2008).
Structural evidence for substrate-induced synergism and half-sites reactivity in biotin carboxylase.
|
| |
Protein Sci,
17,
1706-1718.
|
 |
|
PDB codes:
|
 |
|
|
|
|
|
 |
R.Mosca,
and
T.R.Schneider
(2008).
RAPIDO: a web server for the alignment of protein structures in the presence of conformational changes.
|
| |
Nucleic Acids Res,
36,
W42-W46.
|
 |
|
|
|
|
 |
S.Jitrapakdee,
M.St Maurice,
I.Rayment,
W.W.Cleland,
J.C.Wallace,
and
P.V.Attwood
(2008).
Structure, mechanism and regulation of pyruvate carboxylase.
|
| |
Biochem J,
413,
369-387.
|
 |
|
|
|
|
 |
S.O.Nilsson Lill,
J.Gao,
and
G.L.Waldrop
(2008).
Molecular dynamics simulations of biotin carboxylase.
|
| |
J Phys Chem B,
112,
3149-3156.
|
 |
|
|
|
|
 |
S.Xiang,
and
L.Tong
(2008).
Crystal structures of human and Staphylococcus aureus pyruvate carboxylase and molecular insights into the carboxyltransfer reaction.
|
| |
Nat Struct Mol Biol,
15,
295-302.
|
 |
|
PDB codes:
|
 |
|
|
|
|
|
 |
Y.S.Cho,
J.I.Lee,
D.Shin,
H.T.Kim,
Y.H.Cheon,
C.I.Seo,
Y.E.Kim,
Y.L.Hyun,
Y.S.Lee,
K.Sugiyama,
S.Y.Park,
S.Ro,
J.M.Cho,
T.G.Lee,
and
Y.S.Heo
(2008).
Crystal structure of the biotin carboxylase domain of human acetyl-CoA carboxylase 2.
|
| |
Proteins,
70,
268-272.
|
 |
|
PDB code:
|
 |
|
|
|
|
|
 |
M.St Maurice,
L.Reinhardt,
K.H.Surinya,
P.V.Attwood,
J.C.Wallace,
W.W.Cleland,
and
I.Rayment
(2007).
Domain architecture of pyruvate carboxylase, a biotin-dependent multifunctional enzyme.
|
| |
Science,
317,
1076-1079.
|
 |
|
PDB code:
|
 |
|
|
|
|
|
 |
S.Jitrapakdee,
K.H.Surinya,
A.Adina-Zada,
S.W.Polyak,
C.Stojkoski,
R.Smyth,
G.W.Booker,
W.W.Cleland,
P.V.Attwood,
and
J.C.Wallace
(2007).
Conserved Glu40 and Glu433 of the biotin carboxylase domain of yeast pyruvate carboxylase I isoenzyme are essential for the association of tetramers.
|
| |
Int J Biochem Cell Biol,
39,
2120-2134.
|
 |
|
|
|
|
 |
S.Kondo,
Y.Nakajima,
S.Sugio,
S.Sueda,
M.N.Islam,
and
H.Kondo
(2007).
Structure of the biotin carboxylase domain of pyruvate carboxylase from Bacillus thermodenitrificans.
|
| |
Acta Crystallogr D Biol Crystallogr,
63,
885-890.
|
 |
|
PDB code:
|
 |
|
|
|
|
|
 |
C.H.Pai,
B.Y.Chiang,
T.P.Ko,
C.C.Chou,
C.M.Chong,
F.J.Yen,
S.Chen,
J.K.Coward,
A.H.Wang,
and
C.H.Lin
(2006).
Dual binding sites for translocation catalysis by Escherichia coli glutathionylspermidine synthetase.
|
| |
EMBO J,
25,
5970-5982.
|
 |
|
PDB codes:
|
 |
|
|
|
|
|
 |
L.Tong,
and
H.J.Harwood
(2006).
Acetyl-coenzyme A carboxylases: versatile targets for drug discovery.
|
| |
J Cell Biochem,
99,
1476-1488.
|
 |
|
|
|
|
 |
Y.Shen,
C.Y.Chou,
G.G.Chang,
and
L.Tong
(2006).
Is dimerization required for the catalytic activity of bacterial biotin carboxylase?
|
| |
Mol Cell,
22,
807-818.
|
 |
|
PDB codes:
|
 |
|
|
|
|
|
 |
D.M.Standley,
H.Toh,
and
H.Nakamura
(2005).
GASH: an improved algorithm for maximizing the number of equivalent residues between two protein structures.
|
| |
BMC Bioinformatics,
6,
221.
|
 |
|
|
|
|
 |
K.Izui,
H.Matsumura,
T.Furumoto,
and
Y.Kai
(2004).
Phosphoenolpyruvate carboxylase: a new era of structural biology.
|
| |
Annu Rev Plant Biol,
55,
69-84.
|
 |
|
|
|
|
 |
M.R.Baumgartner,
M.F.Dantas,
T.Suormala,
S.Almashanu,
C.Giunta,
D.Friebel,
B.Gebhardt,
B.Fowler,
G.F.Hoffmann,
E.R.Baumgartner,
and
D.Valle
(2004).
Isolated 3-methylcrotonyl-CoA carboxylase deficiency: evidence for an allele-specific dominant negative effect and responsiveness to biotin therapy.
|
| |
Am J Hum Genet,
75,
790-800.
|
 |
|
|
|
|
 |
R.J.Heath,
and
C.O.Rock
(2004).
Fatty acid biosynthesis as a target for novel antibacterials.
|
| |
Curr Opin Investig Drugs,
5,
146-153.
|
 |
|
|
|
|
 |
S.Kondo,
Y.Nakajima,
S.Sugio,
J.Yong-Biao,
S.Sueda,
and
H.Kondo
(2004).
Structure of the biotin carboxylase subunit of pyruvate carboxylase from Aquifex aeolicus at 2.2 A resolution.
|
| |
Acta Crystallogr D Biol Crystallogr,
60,
486-492.
|
 |
|
PDB code:
|
 |
|
|
|
|
|
 |
S.Sueda,
M.N.Islam,
and
H.Kondo
(2004).
Protein engineering of pyruvate carboxylase: investigation on the function of acetyl-CoA and the quaternary structure.
|
| |
Eur J Biochem,
271,
1391-1400.
|
 |
|
|
|
|
 |
T.Kanamori,
N.Kanou,
H.Atomi,
and
T.Imanaka
(2004).
Enzymatic characterization of a prokaryotic urea carboxylase.
|
| |
J Bacteriol,
186,
2532-2539.
|
 |
|
|
|
|
 |
Y.Shen,
S.L.Volrath,
S.C.Weatherly,
T.D.Elich,
and
L.Tong
(2004).
A mechanism for the potent inhibition of eukaryotic acetyl-coenzyme A carboxylase by soraphen A, a macrocyclic polyketide natural product.
|
| |
Mol Cell,
16,
881-891.
|
 |
|
PDB codes:
|
 |
|
|
|
|
|
 |
J.E.Cronan,
and
G.L.Waldrop
(2002).
Multi-subunit acetyl-CoA carboxylases.
|
| |
Prog Lipid Res,
41,
407-435.
|
 |
|
|
|
|
 |
J.W.Campbell,
and
J.E.Cronan
(2001).
Bacterial fatty acid biosynthesis: targets for antibacterial drug discovery.
|
| |
Annu Rev Microbiol,
55,
305-332.
|
 |
|
|
|
|
 |
L.H.Weaver,
K.Kwon,
D.Beckett,
and
B.W.Matthews
(2001).
Competing protein:protein interactions are proposed to control the biological switch of the E coli biotin repressor.
|
| |
Protein Sci,
10,
2618-2622.
|
 |
|
PDB codes:
|
 |
|
|
|
|
|
 |
M.E.Gallardo,
L.R.Desviat,
J.M.Rodríguez,
J.Esparza-Gordillo,
C.Pérez-Cerdá,
B.Pérez,
P.Rodríguez-Pombo,
O.Criado,
R.Sanz,
D.H.Morton,
K.M.Gibson,
T.P.Le,
A.Ribes,
S.R.de Córdoba,
M.Ugarte,
and
M.A.Peñalva
(2001).
The molecular basis of 3-methylcrotonylglycinuria, a disorder of leucine catabolism.
|
| |
Am J Hum Genet,
68,
334-346.
|
 |
|
|
|
|
 |
M.R.Baumgartner,
S.Almashanu,
T.Suormala,
C.Obie,
R.N.Cole,
S.Packman,
E.R.Baumgartner,
and
D.Valle
(2001).
The molecular basis of human 3-methylcrotonyl-CoA carboxylase deficiency.
|
| |
J Clin Invest,
107,
495-504.
|
 |
|
|
|
|
 |
R.J.Heath,
S.W.White,
and
C.O.Rock
(2001).
Lipid biosynthesis as a target for antibacterial agents.
|
| |
Prog Lipid Res,
40,
467-497.
|
 |
|
 |
 |
|
The most recent references are shown first.
Citation data come partly from CiteXplore and partly
from an automated harvesting procedure. Note that this is likely to be
only a partial list as not all journals are covered by
either method. However, we are continually building up the citation data
so more and more references will be included with time.
Where a reference describes a PDB structure, the PDB
code is
shown on the right.
|
 |

