 |
PDBsum entry 1cvh

|

|

|
|
 |
Contents |
 |
|
|

|

|
|
|
|
|
|
|
|
* Residue conservation analysis
|
|
|
|
 |
|
|
 |
 |
 |
 |
Enzyme class 2:
|
 |
E.C.4.2.1.1
- carbonic anhydrase.
|
|
 |
 |
 |
 |
 |
Reaction:
|
 |
hydrogencarbonate + H+ = CO2 + H2O
|
 |
 |
 |
 |
 |
 hydrogencarbonate
hydrogencarbonate
|
+
|
H(+)
|
=
|
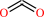 CO2
CO2
|
+
|
H2O
|
|
 |
 |
 |
 |
 |
 |
 |
 |
Cofactor:
|
 |
Zn(2+)
|
 |
 |
 |
 |
 |
Enzyme class 3:
|
 |
E.C.4.2.1.69
- cyanamide hydratase.
|
|
 |
 |
 |
 |
 |
Reaction:
|
 |
urea = cyanamide + H2O
|
 |
 |
 |
 |
 |
 urea
urea
|
=
|
 cyanamide
cyanamide
|
+
|
H2O
|
|
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
|
Note, where more than one E.C. class is given (as above), each may
correspond to a different protein domain or, in the case of polyprotein
precursors, to a different mature protein.
|
|
 |
|
Molecule diagrams generated from .mol files obtained from the
KEGG ftp site
|
|
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |

|

|

|
| |
|
|
| |
|
DOI no:
|
Biochemistry
33:15241-15249
(1994)
|
|
PubMed id:
|
|
|
|

|
| |
|
Structural consequences of redesigning a protein-zinc binding site.
|
|
J.A.Ippolito,
D.W.Christianson.
|
|
|

|
| |
ABSTRACT
|
|

|
| |

|
In order to probe the structural importance of zinc ligands in the active site
of human carbonic anhydrase II (CAII), we have determined the three-dimensional
structures of H94C (in metal-bound form), H94C-BME (i.e., disulfide-linked with
beta-mercaptoethanol), H94A, H96C, H119C, and H119D variants of CAII by X-ray
crystallographic methods at resolutions of 2.2, 2.35, 2.25, 2.3, 2.2, and 2.25
A, respectively. Each variant crystallizes isomorphously with the wild-type
enzyme, in which zinc is tetrahedrally coordinated by H94, H96, H119, and
hydroxide ion. The structure of H94C CAII reveals the successful substitution of
the naturally occurring histidine zinc ligand by a cysteine thiolate, and metal
coordination by C94 is facilitated by the plastic structural response of the
beta-sheet superstructure. Importantly, the resulting structure represents the
catalytically active form of the enzyme reported previously [Alexander, R. S.,
Kiefer, L. L., Fierke, C. A., & Christianson, D. W. (1993) Biochemistry 32,
1510-1518]. Contrastingly, the structure of H96C CAII reveals that the
engineered side chain does not coordinate to zinc; instead, zinc is
tetrahedrally liganded by H94, H119, and two solvent molecules. Thus, the
beta-sheet superstructure is not sufficiently plastic in this location to allow
C96 to coordinate to the metal ion. Substitution of the thiolate or carboxylate
group for wild-type histidine in H119C and H119D CAIIs reveals that tetrahedral
metal coordination is maintained in each variant; however, since there is no
plastic structural response of the corresponding beta-strand, a longer
metal-ligand separation results.(ABSTRACT TRUNCATED AT 250 WORDS)

|

|
|
|

|

|
|
 |
 |
|
 |
 |
 |
 |
 |
 |
 |
 |
 |
|
Literature references that cite this PDB file's key reference
|
|
 |
| |
PubMed id
|
 |
Reference
|
 |
|
|
|
 |
A.Tamilselvi,
and
G.Mugesh
(2008).
Zinc and antibiotic resistance: metallo-beta-lactamases and their synthetic analogues.
|
| |
J Biol Inorg Chem,
13,
1039-1053.
|
 |
|
|
|
|
 |
V.M.Krishnamurthy,
G.K.Kaufman,
A.R.Urbach,
I.Gitlin,
K.L.Gudiksen,
D.B.Weibel,
and
G.M.Whitesides
(2008).
Carbonic anhydrase as a model for biophysical and physical-organic studies of proteins and protein-ligand binding.
|
| |
Chem Rev,
108,
946.
|
 |
|
|
|
|
 |
S.Schenk,
J.Notni,
U.Köhn,
K.Wermann,
and
E.Anders
(2006).
Carbon dioxide and related heterocumulenes at zinc and lithium cations: bioinspired reactions and principles.
|
| |
Dalton Trans,
(),
4191-4206.
|
 |
|
|
|
|
 |
A.Hudmon,
and
H.Schulman
(2002).
Neuronal CA2+/calmodulin-dependent protein kinase II: the role of structure and autoregulation in cellular function.
|
| |
Annu Rev Biochem,
71,
473-510.
|
 |
|
|
|
|
 |
I.Lengyel,
S.Fieuw-Makaroff,
A.L.Hall,
A.T.Sim,
J.A.Rostas,
and
P.R.Dunkley
(2000).
Modulation of the phosphorylation and activity of calcium/calmodulin-dependent protein kinase II by zinc.
|
| |
J Neurochem,
75,
594-605.
|
 |
|
|
|
|
 |
D.W.Christianson,
and
J.D.Cox
(1999).
Catalysis by metal-activated hydroxide in zinc and manganese metalloenzymes.
|
| |
Annu Rev Biochem,
68,
33-57.
|
 |
|
|
|
|
 |
J.E.Coleman
(1998).
Zinc enzymes.
|
| |
Curr Opin Chem Biol,
2,
222-234.
|
 |
|
|
|
|
 |
J.A.Hunt,
and
C.A.Fierke
(1997).
Selection of carbonic anhydrase variants displayed on phage. Aromatic residues in zinc binding site enhance metal affinity and equilibration kinetics.
|
| |
J Biol Chem,
272,
20364-20372.
|
 |
|
|
|
|
 |
J.Guo,
and
D.P.Giedroc
(1997).
Zinc site redesign in T4 gene 32 protein: structure and stability of cobalt(II) complexes formed by wild-type and metal ligand substitution mutants.
|
| |
Biochemistry,
36,
730-742.
|
 |
|
|
|
|
 |
L.M.Watkins,
J.M.Kuo,
M.Chen-Goodspeed,
and
F.M.Raushel
(1997).
A combinatorial library for the binuclear metal center of bacterial phosphotriesterase.
|
| |
Proteins,
29,
553-561.
|
 |
|
|
|
|
 |
S.Lindskog
(1997).
Structure and mechanism of carbonic anhydrase.
|
| |
Pharmacol Ther,
74,
1.
|
 |
|
|
|
|
 |
C.C.Huang,,
C.A.Lesburg,
L.L.Kiefer,
C.A.Fierke,
and
D.W.Christianson
(1996).
Reversal of the hydrogen bond to zinc ligand histidine-119 dramatically diminishes catalysis and enhances metal equilibration kinetics in carbonic anhydrase II.
|
| |
Biochemistry,
35,
3439-3446.
|
 |
|
PDB codes:
|
 |
|
|
|
|
|
 |
K.Thirstrup,
C.E.Elling,
S.A.Hjorth,
and
T.W.Schwartz
(1996).
Construction of a high affinity zinc switch in the kappa-opioid receptor.
|
| |
J Biol Chem,
271,
7875-7878.
|
 |
|
|
|
|
 |
L.S.Brinen,
W.S.Willett,
C.S.Craik,
and
R.J.Fletterick
(1996).
X-ray structures of a designed binding site in trypsin show metal-dependent geometry.
|
| |
Biochemistry,
35,
5999-6009.
|
 |
|
PDB codes:
|
 |
|
|
|
|
|
 |
D.Barrick
(1995).
Depletion and replacement of protein metal ligands.
|
| |
Curr Opin Biotechnol,
6,
411-418.
|
 |
|
 |
 |
|
The most recent references are shown first.
Citation data come partly from CiteXplore and partly
from an automated harvesting procedure. Note that this is likely to be
only a partial list as not all journals are covered by
either method. However, we are continually building up the citation data
so more and more references will be included with time.
Where a reference describes a PDB structure, the PDB
codes are
shown on the right.
|
 |

