 |
PDBsum entry 1ce8

|

|

|
|
 |
Contents |
 |
|
|

|

|
|
|
|
|
|
|
|
|
|
* Residue conservation analysis
|
|
|

|
|
PDB id:
|
 |
|
 |
| Name: |
 |
Ligase imp
|
 |
|
Title:
|
 |
Carbamoyl phosphate synthetase from escherichis coli with complexed with the allosteric ligand imp
|
|
Structure:
|
 |
Protein (carbamoyl-phosphate synthase). Chain: a, c, e, g. Engineered: yes. Other_details: long chain. Protein (carbamoyl-phosphate synthase). Chain: b, d, f, h. Engineered: yes. Other_details: short chain
|
|
Source:
|
 |
Escherichia coli. Organism_taxid: 562. Expressed in: escherichia coli. Expression_system_taxid: 562. Expression_system_taxid: 562
|
|
Biol. unit:
|
 |
 Hetero-Tetramer (from PDB file)
Hetero-Tetramer (from PDB file)
|
|
Resolution:
|
 |
|
2.10Å
|
R-factor:
|
0.192
|
R-free:
|
0.250
|
|
|
Authors:
|
 |
J.B.Thoden,F.M.Raushel,H.M.Holden
|
Key ref:

|
 |
J.B.Thoden
et al.
(1999).
The binding of inosine monophosphate to Escherichia coli carbamoyl phosphate synthetase.
J Biol Chem,
274,
22502-22507.
PubMed id:
DOI:

|
 |
|
Date:
|
 |
|
18-Mar-99
|
Release date:
|
26-Jul-99
|
|
|

|

|
|
PROCHECK
|

|

|
|
|
Headers
|
 |
|
|
References
|
|
|
|
|
 |
|
|
 |
 |
 |
 |
Enzyme class:
|
 |
Chains A, B, C, D, E, F, G, H:
E.C.6.3.5.5
- carbamoyl-phosphate synthase (glutamine-hydrolyzing).
|
|
 |
 |
 |
 |
 |

Pathway:
|
 |
Pyrimidine Biosynthesis
|
 |
 |
 |
 |
 |
Reaction:
|
 |
hydrogencarbonate + L-glutamine + 2 ATP + H2O = carbamoyl phosphate + L-glutamate + 2 ADP + phosphate + 2 H+
|
 |
 |
 |
 |
 |
 hydrogencarbonate
hydrogencarbonate
|
+
|
 L-glutamine
L-glutamine
|
+
|
 2
×
ATP
2
×
ATP
|
+
|
H2O
Bound ligand (Het Group name = )
matches with 90.00% similarity
|
=
|
carbamoyl phosphate
Bound ligand (Het Group name = )
corresponds exactly
|
+
|
 L-glutamate
L-glutamate
|
+
|
 2
×
ADP
2
×
ADP
|
+
|
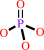 phosphate
phosphate
|
+
|
2
×
H(+)
Bound ligand (Het Group name = )
corresponds exactly
|
|
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
|
Molecule diagrams generated from .mol files obtained from the
KEGG ftp site
|
|
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |

|

|

|
| |
|
|
| |
|
DOI no:
|
J Biol Chem
274:22502-22507
(1999)
|
|
PubMed id:
|
|
|
|

|
| |
|
The binding of inosine monophosphate to Escherichia coli carbamoyl phosphate synthetase.
|
|
J.B.Thoden,
F.M.Raushel,
G.Wesenberg,
H.M.Holden.
|
|
|

|
| |
ABSTRACT
|
|

|
| |

|
Carbamoyl phosphate synthetase (CPS) from Escherichia coli catalyzes the
formation of carbamoyl phosphate, which is subsequently employed in both the
pyrimidine and arginine biosynthetic pathways. The reaction mechanism is known
to proceed through at least three highly reactive intermediates: ammonia,
carboxyphosphate, and carbamate. In keeping with the fact that the product of
CPS is utilized in two competing metabolic pathways, the enzyme is highly
regulated by a variety of effector molecules including potassium and ornithine,
which function as activators, and UMP, which acts as an inhibitor. IMP is also
known to bind to CPS but the actual effect of this ligand on the activity of the
enzyme is dependent upon both temperature and assay conditions. Here we describe
the three-dimensional architecture of CPS with bound IMP determined and refined
to 2.1 A resolution. The nucleotide is situated at the C-terminal portion of a
five-stranded parallel beta-sheet in the allosteric domain formed by Ser(937) to
Lys(1073). Those amino acid side chains responsible for anchoring the nucleotide
to the polypeptide chain include Lys(954), Thr(974), Thr(977), Lys(993),
Asn(1015), and Thr(1017). A series of hydrogen bonds connect the IMP-binding
pocket to the active site of the large subunit known to function in the
phosphorylation of the unstable intermediate, carbamate. This structural
analysis reveals, for the first time, the detailed manner in which CPS
accommodates nucleotide monophosphate effector molecules within the allosteric
domain.

|

|
|
|

|
| |
Selected figure(s)
|
|

|
| |
 |
 |

|
 |

|
 |
Figure 3.
Fig. 3. The IMP binding pocket. Panel a, a close-up view
of those amino acid residues located within approximately 5
Å of atoms of the IMP. Panel b, a cartoon of potential
hydrogen bonds between the ligand and the protein, as indicated
by the dashed lines.
|
 |
Figure 4.
Fig. 4. Superposition of the CPS allosteric binding
pockets with bound IMP or glutamine/inorganic phosphate. The
model described in this report is shown in black, whereas the
structure of CPS complexed with glutamine and inorganic
phosphate, as described by Thoden et al. (2), is displayed in
red. Note the nearly exact correspondence between the inorganic
phosphate and the phosphoryl moiety of the nucleotide.
|
 |
|
|

|
| |
The above figures are
reprinted
by permission from the ASBMB:
J Biol Chem
(1999,
274,
22502-22507)
copyright 1999.
|
|
| |
Figures were
selected
by an automated process.
|
|

|

|
|
 |
 |
|
 |
 |
 |
 |
 |
 |
 |
 |
 |
|
Literature references that cite this PDB file's key reference
|
|
 |
| |
PubMed id
|
 |
Reference
|
 |
|
|
|
 |
C.A.Bottoms,
and
D.Xu
(2008).
Wanted: unique names for unique atom positions. PDB-wide analysis of diastereotopic atom names of small molecules containing diphosphate.
|
| |
BMC Bioinformatics,
9,
S16.
|
 |
|
|
|
|
 |
J.L.Johnson,
J.K.West,
A.D.Nelson,
and
G.D.Reinhart
(2007).
Resolving the fluorescence response of Escherichia coli carbamoyl phosphate synthetase: mapping intra- and intersubunit conformational changes.
|
| |
Biochemistry,
46,
387-397.
|
 |
|
|
|
|
 |
J.B.Thoden,
X.Huang,
J.Kim,
F.M.Raushel,
and
H.M.Holden
(2004).
Long-range allosteric transitions in carbamoyl phosphate synthetase.
|
| |
Protein Sci,
13,
2398-2405.
|
 |
|
PDB code:
|
 |
|
|
|
|
|
 |
V.Serre,
B.Penverne,
J.L.Souciet,
S.Potier,
H.Guy,
D.Evans,
P.Vicart,
and
G.Hervé
(2004).
Integrated allosteric regulation in the S. cerevisiae carbamylphosphate synthetase - aspartate transcarbamylase multifunctional protein.
|
| |
BMC Biochem,
5,
6.
|
 |
|
|
|
|
 |
M.M.Horvath,
and
N.V.Grishin
(2001).
The C-terminal domain of HPII catalase is a member of the type I glutamine amidotransferase superfamily.
|
| |
Proteins,
42,
230-236.
|
 |
|
 |
 |
|
The most recent references are shown first.
Citation data come partly from CiteXplore and partly
from an automated harvesting procedure. Note that this is likely to be
only a partial list as not all journals are covered by
either method. However, we are continually building up the citation data
so more and more references will be included with time.
Where a reference describes a PDB structure, the PDB
code is
shown on the right.
|
 |
|

