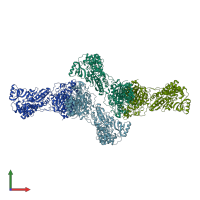
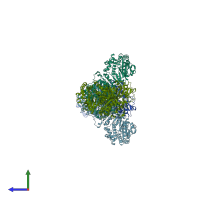
Function and Biology Details
Reactions catalysed:
(3R)-3-hydroxyacyl-[acyl-carrier-protein] + NADP(+) = 3-oxoacyl-[acyl-carrier-protein] + NADPH
An acyl-[acyl-carrier protein] + NADP(+) = a trans-2,3-dehydroacyl-[acyl-carrier protein] + NADPH
Acetyl-CoA + [acyl-carrier-protein] = CoA + acetyl-[acyl-carrier-protein]
Malonyl-CoA + an [acyl-carrier-protein] = CoA + a malonyl-[acyl-carrier-protein]
Acyl-[acyl-carrier-protein] + malonyl-[acyl-carrier-protein] = 3-oxoacyl-[acyl-carrier-protein] + CO(2) + [acyl-carrier-protein]
Acetyl-CoA + n malonyl-CoA + 2n NADPH = a long-chain fatty acid + (n+1) CoA + n CO(2) + 2n NADP(+)
Oleoyl-[acyl-carrier-protein] + H(2)O = [acyl-carrier-protein] + oleate
A (3R)-3-hydroxyacyl-[acyl-carrier protein] = a trans-2-enoyl-[acyl-carrier protein] + H(2)O
Biochemical function:
Biological process:
Cellular component:
- not assigned
Sequence domains:
- Acyl transferase
- Polyketide synthase, beta-ketoacyl synthase domain
- Thiolase-like
- Polyketide synthase, dehydratase domain
- Polyketide synthase, dehydratase domain superfamily
- Malonyl-CoA ACP transacylase, ACP-binding
- Acyl transferase/acyl hydrolase/lysophospholipase
- Acyl transferase domain superfamily
4 more domains
Structure analysis Details
Assembly composition:
homo dimer (preferred)
Assembly name:
Fatty acid synthase (preferred)
PDBe Complex ID:
PDB-CPX-155926 (preferred)
Entry contents:
1 distinct polypeptide molecule
Macromolecule: